
Concept explainers
(a)
Interpretation: The expected molecular ion for the given compound is to be determined.
Concept introduction: Molecular mass is the sum of the atomic weights of each constituent element multiplied by the number of atoms of that element. The molecular weight of compound is equal to the mass of the molecular ion.
Answer to Problem 13.25P
The structure of the given compound is,
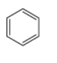
The expected molecular ion peak of
Explanation of Solution
The molecular formula of given compound is
The
The molecular weight of the compound is equal to the mass of the molecular ion.
Hence, the expected molecular ion peak of
The expected molecular ion peak of
(b)
Interpretation: The expected molecular ion for the given compound is to be determined.
Concept introduction: Molecular mass is the sum of the atomic weights of each constituent element multiplied by the number of atoms of that element. The molecular weight of compound is equal to the mass of the molecular ion.
Answer to Problem 13.25P
The structure of the given compound is,
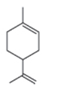
The expected molecular ion peak of
Explanation of Solution
The molecular formula of given compound is
The
The molecular weight of the compound is equal to the mass of the molecular ion.
Hence, the expected molecular ion peak of
The expected molecular ion peak of
(c)
Interpretation: The expected molecular ion for the given compound is to be determined.
Concept introduction: Molecular mass is the sum of the atomic weights of each constituent element multiplied by the number of atoms of that element. The molecular weight of compound is equal to the mass of the molecular ion.
Answer to Problem 13.25P
The structure of the given compound is,
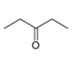
The expected molecular ion peak of
Explanation of Solution
The molecular formula of given compound is
The
The molecular weight of the compound is equal to the mass of the molecular ion.
The expected molecular ion peak of
The expected molecular ion peak of
(d)
Interpretation: The expected molecular ion for the given compound is to be determined.
Concept introduction: Molecular mass is the sum of the atomic weights of each constituent element multiplied by the number of atoms of that element. The molecular weight of compound is equal to the mass of the molecular ion.
Answer to Problem 13.25P
The structure of the given compound is,
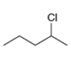
The larger molecular ion
Explanation of Solution
The molecular formula of given compound is
The
The
The molecular weight of the compound is equal to the mass of the molecular ion.
Hence, the larger molecular ion
The larger molecular ion
(e)
Interpretation: The expected molecular ion for the given compound is to be determined.
Concept introduction: Molecular mass is the sum of the atomic weights of each constituent element multiplied by the number of atoms of that element. The molecular weight of compound is equal to the mass of the molecular ion.
Answer to Problem 13.25P
The structure of the given compound is,
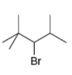
The larger molecular ion
Explanation of Solution
The molecular formula of given compound is
The
The
The molecular weight of the compound is equal to the mass of the molecular ion.
Hence, the larger molecular ion
The larger molecular ion
Want to see more full solutions like this?
Chapter 13 Solutions
Organic Chemistry
- d. b. e o C. a. O 6. Draw at least 3 additional (l.e., in addition to the structure provided) valid resonance (major contributors only) forms of the following ions: Ⓒarrow_forwardMCQ 175: A fluorine atom (F) affects pair of an electron in a covalent bond, which is A. weakest B. strongest C. neutral D. constantarrow_forwardProblem 4.73 Identify bonds polar non polararrow_forward
- A. Choose the letter of the best answer. 1. What is a saturated hydrocarbon? A. Any compound consisting of carbon and hydrogen only. B. Any compound consisting of carbon and hydrogen and oxygen only. C. Any compound consisting of carbon and hydrogen only, in which some of the carbon atoms are joined to each other by double or triple bonds. D. Any compound consisting of carbon and hydrogen only, in which all the carbon atoms are joined to each other by single bonds. 2. What kind of hydrocarbon is C3H14? A. alkane C. alkyne B. alkene D. alcohol H. H H. 3. This molecule is an example of an... H. A. alkane C. alkyne B. alkene D. alcohol 4. Which is an example of an ether? А. CН3ОН B. CH3CH2CH2CI C. CH3CH2OCH2CH3 D. CH3CH2COOCH3 5. What is the simplest organic compound? A. methane C. ethanol B. acetone D. ethanearrow_forwardWhich of the following carbon to carbon bonds has the shortest bond? H. H. H. C=C - C-H - H. H H. H. Option 1 Option 2 Option 3 either A or Barrow_forwardWhich is the strongest bond? A. B. D. E.arrow_forward
- Predict the relative bond lengths of the three carbon–oxygen bonds in the carbonate ion. a. What is the charge on each oxygen?arrow_forwardFollow the curved arrows to draw a second resonance structure for each species. a. b. :o: C. -NEN: NH₂arrow_forwardb)Answer the following for the structure given below. H. H. b(i) no of bond pair electrons b(ii) no of lone pair electrons b(iii) no of single bond b(iv) no of double bondarrow_forward
- O REPRESENTATIONS OF ORGANIC MOLECULES Identifying isomers and resonance structures Determine the relationship between Structure A and Structure B in each row of the table. Structure A Structure B Relationship O isomers :0: HH H H H. H H. O resonance structures H. H. H. H. O neither H O Isomers H :0: H H H C-H H- H ö=C -H- O resonance structures .. H. H. o neither H. O isomers H. H-N=c=N-c-H -NEN: O resonance structures H H H. O neither w DELLarrow_forwardWrite a dash, condensed and bond-line structural formulas of each compound given below. Structural Formula Ball-and-stick Model Dash Condensed Bond-line a. b. C. d. *Note: White = H, Black = C and Red = Oarrow_forward3. What molecular ion is expected for the following compounds? a. b. Br d. j CIarrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

