
(a)
Interpretation:
The missing reactant in the given conversion has to be identified.
Concept Introduction:
Alkylation:
Benzene undergoes alkylation reaction in presence of

Halogenation:
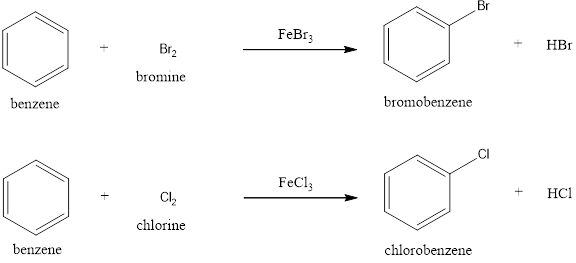
Benzene undergoes halogenation reaction in presence of
(b)
Interpretation:
The missing product in the given conversion has to be identified.
Concept Introduction:
Aromatic hydrocarbons undergo substitution reactions rather than addition reactions. Even though the aromatic hydrocarbon has double bonds in its structure, they undergo substitution reaction only because the double bonds are involved in delocalized bonding that is present in the ring system.
Alkylation:
Benzene undergoes alkylation reaction in presence of

Halogenation:
Benzene undergoes halogenation reaction in presence of

(c)
Interpretation:
The missing reactant in the given conversion has to be identified.
Concept Introduction:
Aromatic hydrocarbons undergo substitution reactions rather than addition reactions. Even though the aromatic hydrocarbon has double bonds in its structure, they undergo substitution reaction only because the double bonds are involved in delocalized bonding that is present in the ring system.
Alkylation:
Benzene undergoes alkylation reaction in presence of

Halogenation:
Benzene undergoes halogenation reaction in presence of

Trending nowThis is a popular solution!

Chapter 13 Solutions
General, Organic, and Biological Chemistry
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,

