
Concept explainers
Assign each of the compounds in Problem 13-108 an IUPAC name in which the substituents on the benzene ring are located using the ortho-, meta-, para-prefix system.
(a)
Interpretation:
The IUPAC name has to be given for the disubstituted benzene derivative by using ortho-, meta-, para- prefix system.
Concept Introduction:
When hydrogen atoms are replaced by one or more groups in benzene is known as substitution reaction and the compounds produced is benzene derivatives.
Benzene derivative with one substituent:
IUPAC system of naming monosubstituted benzene derivatives uses the name of substituent as prefix to the name benzene. If the group that is present in benzene cannot be named easily means, then the benzene ring is often treated as group attached to this substituent. The benzene ring is known as phenyl in this approach.
Benzene derivative with two substituents:
When benzene ring contains two substituents it is known as disubstituted benzene derivative. Three isomers are possible for the disubstituted benzene derivative. The prefix used in IUPAC name are,
Ortho- means disubstitution in 1,2
Meta- means disubstitution in 1,3
Para- means disubstitution in 1,4
When both the substituents present on the benzene ring imparts a special name, where all the substituents are cited in alphabetical order before the ending –benzene. The carbon that bears the group with alphabetical priority is given number 1.
Benzene derivatives with three or more substituents:
More than two groups are present in the benzene ring means, their positions are numbered. The numbering is always done in a way that the carbon atom bearing substituent gets the lowest numbering possible. If there is a choice of numbering system, then the group that comes alphabetically first is given the lowest number.
Answer to Problem 13.110EP
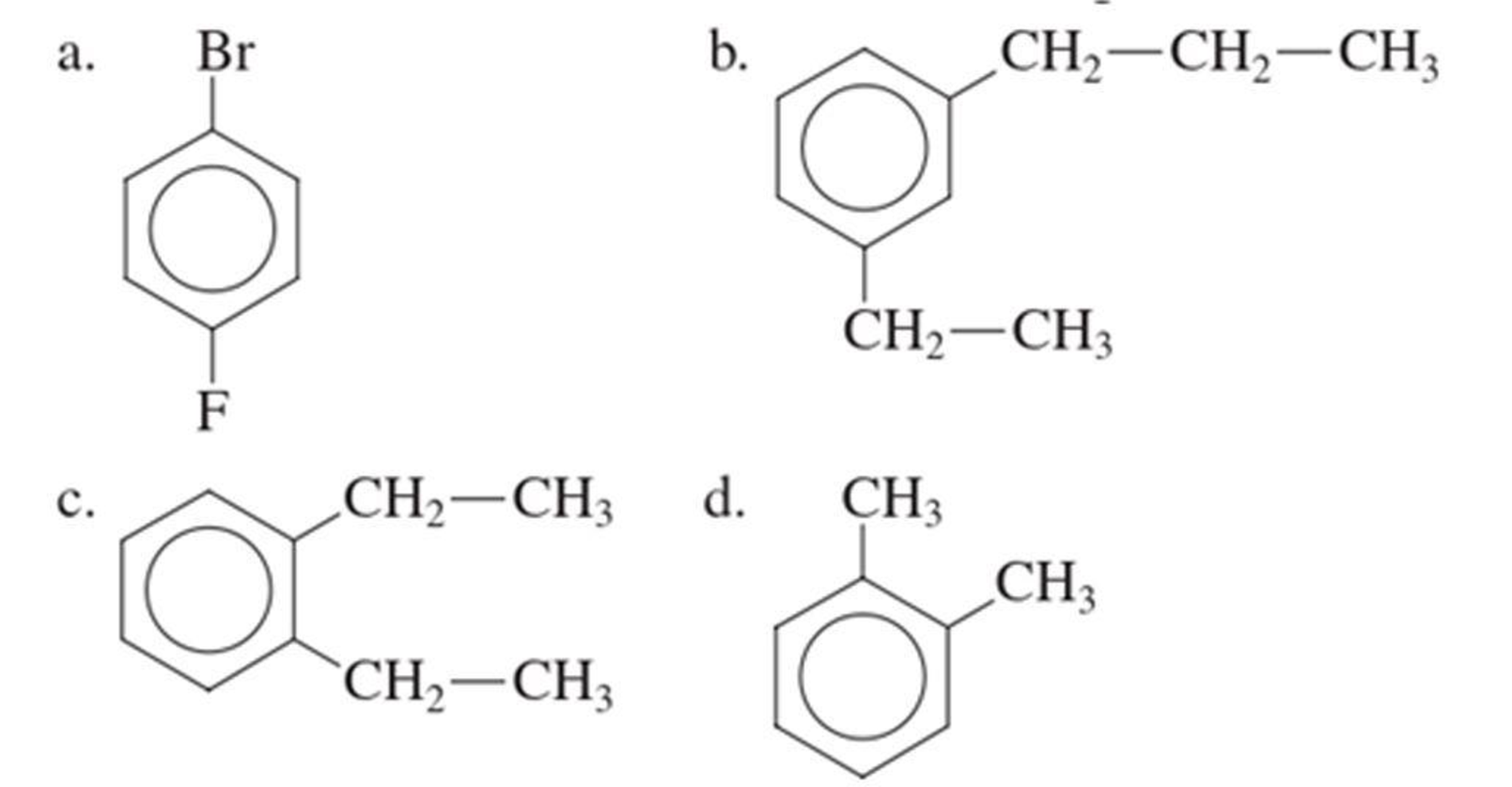
The IUPAC name of the given compound is p-bromofluorobenzene.
Explanation of Solution
Given structure is,

From the structure given above, it is found a fluorine atom and a bromine atom are present as substituent. The numbering has to be given so that the substituents get the least numbering. The numbering has to be given considering the alphabetical order of the substituents. This puts the bromine in number 1 and fluorine in number 4. As the substituents are present in first and fourth position, prefix para- can be used to name the compound. Therefore, the IUPAC name can be given as p-bromofluorobenzene.
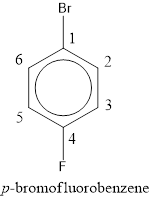
The IUPAC name can be given as p-bromofluorobenzene.
The correct IUPAC name for the given compound is given.
(b)
Interpretation:
The IUPAC name has to be given for the disubstituted benzene derivative by using ortho-, meta-, para- prefix system.
Concept Introduction:
When hydrogen atoms are replaced by one or more groups in benzene is known as substitution reaction and the compounds produced is benzene derivatives.
Benzene derivative with one substituent:
IUPAC system of naming monosubstituted benzene derivatives uses the name of substituent as prefix to the name benzene. If the group that is present in benzene cannot be named easily means, then the benzene ring is often treated as group attached to this substituent. The benzene ring is known as phenyl in this approach.
Benzene derivative with two substituents:
When benzene ring contains two substituents it is known as disubstituted benzene derivative. Three isomers are possible for the disubstituted benzene derivative. The prefix used in IUPAC name are,
Ortho- means disubstitution in 1,2
Meta- means disubstitution in 1,3
Para- means disubstitution in 1,4
When both the substituents present on the benzene ring imparts a special name, where all the substituents are cited in alphabetical order before the ending –benzene. The carbon that bears the group with alphabetical priority is given number 1.
Benzene derivatives with three or more substituents:
More than two groups are present in the benzene ring means, their positions are numbered. The numbering is always done in a way that the carbon atom bearing substituent gets the lowest numbering possible. If there is a choice of numbering system, then the group that comes alphabetically first is given the lowest number.
Answer to Problem 13.110EP
The IUPAC name of the given compound is m-ethylpropylbenzene.
Explanation of Solution
Given structure is,
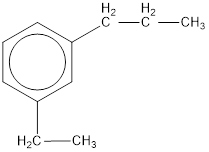
From the structure given above, it is found an ethyl group and a propyl group is present as substituents. The numbering has to be given so that the substituents get the least numbering. The numbering has to be given considering the alphabetical order of the substituents. This puts the ethyl in number 1 and propyl in number 3. As the susbtituents are present in first and third carbon atom, meta- can be used in the name as prefix. Therefore, the IUPAC name can be given as m-ethylpropylbenzene.
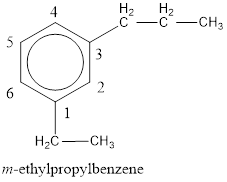
The IUPAC name can be given as m-ethylpropylbenzene.
The correct IUPAC name for the given compound is given.
(c)
Interpretation:
The IUPAC name has to be given for the disubstituted benzene derivative by using ortho-, meta-, para- prefix system.
Concept Introduction:
When hydrogen atoms are replaced by one or more groups in benzene is known as substitution reaction and the compounds produced is benzene derivatives.
Benzene derivative with one substituent:
IUPAC system of naming monosubstituted benzene derivatives uses the name of substituent as prefix to the name benzene. If the group that is present in benzene cannot be named easily means, then the benzene ring is often treated as group attached to this substituent. The benzene ring is known as phenyl in this approach.
Benzene derivative with two substituents:
When benzene ring contains two substituents it is known as disubstituted benzene derivative. Three isomers are possible for the disubstituted benzene derivative. The prefix used in IUPAC name are,
Ortho- means disubstitution in 1,2
Meta- means disubstitution in 1,3
Para- means disubstitution in 1,4
When both the substituents present on the benzene ring imparts a special name, where all the substituents are cited in alphabetical order before the ending –benzene. The carbon that bears the group with alphabetical priority is given number 1.
Benzene derivatives with three or more substituents:
More than two groups are present in the benzene ring means, their positions are numbered. The numbering is always done in a way that the carbon atom bearing substituent gets the lowest numbering possible. If there is a choice of numbering system, then the group that comes alphabetically first is given the lowest number.
Answer to Problem 13.110EP
The IUPAC name of the given compound is o-diethylbenzene.
Explanation of Solution
Given structure is,
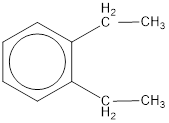
From the structure given above, it is found two ethyl groups are present as substituent. The numbering has to be given so that the substituents get the least numbering. This puts one ethyl group in position 1 and another ethyl group in position 2. As the groups substituted are identical, alphabetical order does not play a part but prefix di- has to be added. The groups that are substituted is present in first and second carbon atom. Hence, prefix ortho- can be used to name the compound. Therefore, the IUPAC name can be given as o-diethylbenzene.
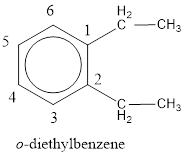
The IUPAC name can be given as o-diethylbenzene.
The correct IUPAC name for the given compound is given.
(d)
Interpretation:
The IUPAC name has to be given for the disubstituted benzene derivative by using ortho-, meta-, para- prefix system.
Concept Introduction:
When hydrogen atoms are replaced by one or more groups in benzene is known as substitution reaction and the compounds produced is benzene derivatives.
Benzene derivative with one substituent:
IUPAC system of naming monosubstituted benzene derivatives uses the name of substituent as prefix to the name benzene. If the group that is present in benzene cannot be named easily means, then the benzene ring is often treated as group attached to this substituent. The benzene ring is known as phenyl in this approach.
Benzene derivative with two substituents:
When benzene ring contains two substituents it is known as disubstituted benzene derivative. Three isomers are possible for the disubstituted benzene derivative. The prefix used in IUPAC name are,
Ortho- means disubstitution in 1,2
Meta- means disubstitution in 1,3
Para- means disubstitution in 1,4
When both the substituents present on the benzene ring imparts a special name, where all the substituents are cited in alphabetical order before the ending –benzene. The carbon that bears the group with alphabetical priority is given number 1.
Benzene derivatives with three or more substituents:
More than two groups are present in the benzene ring means, their positions are numbered. The numbering is always done in a way that the carbon atom bearing substituent gets the lowest numbering possible. If there is a choice of numbering system, then the group that comes alphabetically first is given the lowest number.
Answer to Problem 13.110EP
The IUPAC name of the given compound is o-dimethylbenzene.
Explanation of Solution
Given structure is,
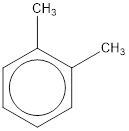
From the structure given above, it is found two methyl groups are present as substituent. The numbering has to be given so that the substituents get the least numbering. This puts one methyl group in position 1 and another methyl group in position 2. As the groups substituted are identical, alphabetical order does not play a part but prefix di- has to be added. The groups that are substituted is present in first and second carbon atom. Hence, prefix ortho- can be used to name the compound. Therefore, the IUPAC name can be given as o-dimethylbenzene.
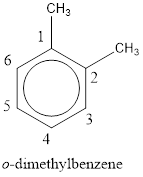
The IUPAC name can be given as o-dimethylbenzene.
The correct IUPAC name for the given compound is given.
Want to see more full solutions like this?
Chapter 13 Solutions
General, Organic, and Biological Chemistry Seventh Edition
- Following are Fischer projections for a group of five-carbon sugars, all of which are aldopentoses. Identify the pairs that are enantiomers and the pairs that are epimers. (The sugars shown here are not all of the possible five-carbon )arrow_forwardFollowing are Fischer projections for a groupof five-carbon sugars, all of which are aldopentoses. Identify thepairs that are enantiomers and the pairs that are epimers. (Thesugars shown here are not all of the possible five-carbon sugars.)arrow_forwardIn the monosaccharide derivatives known as sugar alcohols, the carbonyl oxygen is reduced to a hydroxyl group. For example, D-glyceraldehyde can be reduced to glycerol. However, this sugar alcohol is no longer designated D or L. Why?arrow_forward
- Draw the four stereoisomers of threonine as Fischer projections.arrow_forwardWhich form of lysine in Problem 18.56 is the zwitterion? What is the pI for the zwitterion?arrow_forwardFollowing are Fischer projection for a group of five carbon sugars,all of which are aldopentoses. Identify the pairs that are enantiomers and the pairs that are epimers.(The sugar shown herebare not all of the possible five carbon sugars.)arrow_forward
- Which form of aspartic acid in Problem 18.54 is the zwitterion? What is the pI for the zwitterion?arrow_forwardGentiobiose has the molecular formula C12H22O11 and has been isolated from gentian root and by hydrolysis of amygdalin. Gentiobiose exists in two different forms, one melting at 86°C and the other at 190°C. The lower melting form is dextrorotatory ([α] 16°), the higher melting one is levorotatory ([α] -6°). The rotation of an aqueous solution of either form, however, gradually changes until a final value of [α] 9.6° is observed. Hydrolysis of gentiobiose is efficiently catalyzed by emulsin and produces two moles of D-glucose per mole of gentiobiose. Gentiobiose forms an octamethylether, which on hydrolysis in dilute acid yields 2,3,4,6-tetra-O-methyl-D-glucose and 2,3,4-tri-O-methyl-D-glucose. What is the structure of gentiobiose? EXPLAIN IN DETAIL.arrow_forwardFollowing are Fischer projections for a group of five-carbon sugars, all of which are aldopentoses. Identify the pairs that are enantiomers and the pairs that are epimers. (The sugars shown here are not all of the possible five-carbon sugars.) 1 СНО 2 СНО СНО Н-С—ОН Н-С—ОН Н—С—ОН Н-С—ОН НО —С—Н Н-С—ОН Н- С—ОН НО—С— Н НО—С—Н CH̟OH CH̟OH ČH̟OH в iоchemistry |61 STATE ATION Republic of the Philippines Romblon State University Romblan, Philippines 4 СНО 5 СНО 6 СНО НО—С—Н Н-С—ОН НО -С—Н Н—С—ОН НО -С—Н НО -С—Н H-C–OH Н-С—ОН НО—С—Н CH,OH ČH,OH CH,OHarrow_forward
- why is 2,2 diethylbutane not the correct IUPAC namearrow_forwardComplete the following table by providing the Fischer and Haworth Projections of the given sugars. (Please be careful with the alpha and beta, and the complete and abbreviated) (not a graded question) Sugar Fischer Projection Haworth Projection (Complete) alpha-anomer Haworth Projection (Abbreviated) beta-anomer L-Galactose L-Fructosearrow_forwardWhat is isopropylthiogalactoside (IPTG) ?arrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





