
(a)
Interpretation:
The IUPAC name for the given molecule has to be assigned including the prefix cis- or trans-.
Concept Introduction:
- The suffix –ane has to be replaced with the suffix –ene. This is used to indicate the presence of double bond.
- The longest continuous chain of carbon atoms has to be chosen that contains both carbon atoms of the double bond.
- The parent carbon chain has to be numbered in a way so that the numbering begins at the end near to the double bond. In case if the double bond is equidistant from both ends, then numbering has to be done from the end that is closer to substituents.
- The position of the double bond has to be given a single number which is lower‑numbered carbon atom that is present in the double bond.
- Suffixes like –diene, -triene, -tetrene, and so on are used when the compound contains more than one double bond.
- In case of cycloalkenes which do not have any substitution, the numbering is not needed to locate the double bond because the bond is assumed to be between the carbons 1 and 2.
- In case if substituents are present in cycloalkene, then the double‑bonded carbon
atoms are numbered 1 and 2 in a direction where the substituent gets the lower number. - If the cycloalkenes contain more than one double bond, then one double bond is assigned the numbers 1 and 2 followed by the other double bond so that the lowest number possible is given.
Structural formula where a line represent carbon‑carbon bond and the carbon atom is considered to be present in each point and the end of lines is known as Line-angle structural formula. To indicate a double bond between carbon atom, double line is used.
If stereoisomers are possible for the alkene, the stereoinformation is shown in the IUPAC name by adding prefix cis- or trans- with respect to the groups present on the same side of double bond or opposite side of double bond.
(a)
Answer to Problem 13.49EP
The IUPAC name for the given compound is cis-2-pentene.
Explanation of Solution
Given compound is,
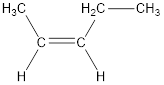
The longest continuous carbon chain present in the given compound is five carbon atoms. Hence, the parent
As the given compound contains a double bond, the suffix –ane is replaced by –ene. Therefore, the name obtained is pentene.
Numbering has to be given in a way that the carbon atoms present in the double bond gets the least numbering. Therefore, the name of the given compound is 2-pentene.
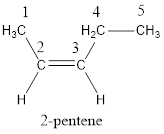
In order to include the stereo information, the groups attached to the double‑bonded carbon atoms are looked into.
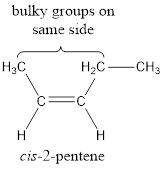
The bulky groups are present on same side of the double bond. Hence, the configuration of the given molecule is cis-. Therefore, the IUPAC name can be given as cis-2-pentene.
IUPAC name for the given molecule is assigned.
(b)
Interpretation:
The IUPAC name for the given molecule has to be assigned including the prefix cis- or trans-.
Concept Introduction:
IUPAC nomenclature for alkene: There are about eight rules to be followed in giving IUPAC name for alkene.
- The suffix –ane has to be replaced with the suffix –ene. This is used to indicate the presence of double bond.
- The longest continuous chain of carbon atoms has to be chosen that contains both carbon atoms of the double bond.
- The parent carbon chain has to be numbered in a way so that the numbering begins at the end near to the double bond. In case if the double bond is equidistant from both ends, then numbering has to be done from the end that is closer to substituents.
- The position of the double bond has to be given a single number which is lower‑numbered carbon atom that is present in the double bond.
- Suffixes like –diene, -triene, -tetrene, and so on are used when the compound contains more than one double bond.
- In case of cycloalkenes which do not have any substitution, the numbering is not needed to locate the double bond because the bond is assumed to be between the carbons 1 and 2.
- In case if substituents are present in cycloalkene, then the double‑bonded carbon atoms are numbered 1 and 2 in a direction where the substituent gets the lower number.
- If the cycloalkenes contain more than one double bond, then one double bond is assigned the numbers 1 and 2 followed by the other double bond so that the lowest number possible is given.
Structural formula where a line represent carbon‑carbon bond and the carbon atom is considered to be present in each point and the end of lines is known as Line-angle structural formula. To indicate a double bond between carbon atom, double line is used.
If stereoisomers are possible for the alkene, the stereoinformation is shown in the IUPAC name by adding prefix cis- or trans- with respect to the groups present on the same side of double bond or opposite side of double bond.
(b)
Answer to Problem 13.49EP
The IUPAC name for the given compound is trans-1-bromo-2-iodoethene.
Explanation of Solution
Given compound is,
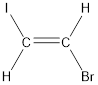
The longest continuous carbon chain present in the given compound is two carbon atoms. Hence, the parent alkane is ethane.
As the given compound contains a double bond, the suffix –ane is replaced by –ene. Therefore, the name obtained is ethene.
Numbering has to be given in a way that the carbon atoms present in the double bond gets the least numbering. This is followed by the substituents present in the given molecule. It is found that an iodine atom is present on the second carbon atom and bromine atom is present on the first carbon atom. Therefore, the name of the given compound is 2-pentene.
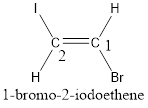
In order to include the stereo information, the groups attached to the double‑bonded carbon atoms are looked into.
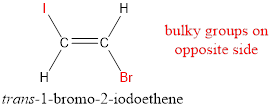
The bulky groups are present on opposite side of the double bond. Hence, the configuration of the given molecule is trans-. Therefore, the IUPAC name can be given as trans-1-bromo-2-iodoethene.
IUPAC name for the given molecule is assigned.
(c)
Interpretation:
The IUPAC name for the given molecule has to be assigned including the prefix cis- or trans-.
Concept Introduction:
IUPAC nomenclature for alkene: There are about eight rules to be followed in giving IUPAC name for alkene.
- The suffix –ane has to be replaced with the suffix –ene. This is used to indicate the presence of double bond.
- The longest continuous chain of carbon atoms has to be chosen that contains both carbon atoms of the double bond.
- The parent carbon chain has to be numbered in a way so that the numbering begins at the end near to the double bond. In case if the double bond is equidistant from both ends, then numbering has to be done from the end that is closer to substituents.
- The position of the double bond has to be given a single number which is lower‑numbered carbon atom that is present in the double bond.
- Suffixes like –diene, -triene, -tetrene, and so on are used when the compound contains more than one double bond.
- In case of cycloalkenes which do not have any substitution, the numbering is not needed to locate the double bond because the bond is assumed to be between the carbons 1 and 2.
- In case if substituents are present in cycloalkene, then the double‑bonded carbon atoms are numbered 1 and 2 in a direction where the substituent gets the lower number.
- If the cycloalkenes contain more than one double bond, then one double bond is assigned the numbers 1 and 2 followed by the other double bond so that the lowest number possible is given.
Structural formula where a line represent carbon‑carbon bond and the carbon atom is considered to be present in each point and the end of lines is known as Line-angle structural formula. To indicate a double bond between carbon atom, double line is used.
If stereoisomers are possible for the alkene, the stereoinformation is shown in the IUPAC name by adding prefix cis- or trans- with respect to the groups present on the same side of double bond or opposite side of double bond.
(c)
Answer to Problem 13.49EP
The IUPAC name for the given compound is tetrafluoroethene.
Explanation of Solution
Given compound is,
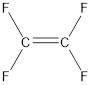
The longest continuous carbon chain present in the given compound is two carbon atoms. Hence, the parent alkane is ethane.
As the given compound contains a double bond, the suffix –ane is replaced by –ene. Therefore, the name obtained is ethene.
In this case, numbering does not make any difference. This is because all the hydrogen atoms are replaced by fluorine atoms. Hence, the IUPAC name can be given as tetrafluoroethene.
Stereoisomers are not possible for the given molecule because same groups are present on the carbon atom that is present on the double bond.
IUPAC name for the given molecule is assigned.
(d)
Interpretation:
The IUPAC name for the given molecule has to be assigned including the prefix cis- or trans-.
Concept Introduction:
IUPAC nomenclature for alkene: There are about eight rules to be followed in giving IUPAC name for alkene.
- The suffix –ane has to be replaced with the suffix –ene. This is used to indicate the presence of double bond.
- The longest continuous chain of carbon atoms has to be chosen that contains both carbon atoms of the double bond.
- The parent carbon chain has to be numbered in a way so that the numbering begins at the end near to the double bond. In case if the double bond is equidistant from both ends, then numbering has to be done from the end that is closer to substituents.
- The position of the double bond has to be given a single number which is lower‑numbered carbon atom that is present in the double bond.
- Suffixes like –diene, -triene, -tetrene, and so on are used when the compound contains more than one double bond.
- In case of cycloalkenes which do not have any substitution, the numbering is not needed to locate the double bond because the bond is assumed to be between the carbons 1 and 2.
- In case if substituents are present in cycloalkene, then the double‑bonded carbon atoms are numbered 1 and 2 in a direction where the substituent gets the lower number.
- If the cycloalkenes contain more than one double bond, then one double bond is assigned the numbers 1 and 2 followed by the other double bond so that the lowest number possible is given.
Structural formula where a line represent carbon‑carbon bond and the carbon atom is considered to be present in each point and the end of lines is known as Line-angle structural formula. To indicate a double bond between carbon atom, double line is used.
If stereoisomers are possible for the alkene, the stereoinformation is shown in the IUPAC name by adding prefix cis- or trans- with respect to the groups present on the same side of double bond or opposite side of double bond.
(d)
Answer to Problem 13.49EP
The IUPAC name for the given compound is 2-methyl-2-butene.
Explanation of Solution
Given compound is,
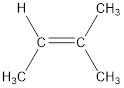
The longest continuous carbon chain present in the given compound is four carbon atoms. Hence, the parent alkane is butane.
As the given compound contains a double bond, the suffix –ane is replaced by –ene. Therefore, the name obtained is butene.
Numbering has to be given in a way that the carbon atoms present in the double bond gets the least numbering. Therefore, the name of the given compound is 2-butene. The substituent present on the longest carbon chain is a methyl group that is present on the second carbon atom. Therefore, the name of the given molecule can be given as,
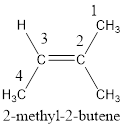
In order to include the stereo information, the groups attached to the double‑bonded carbon atoms are looked into.
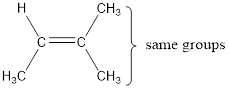
The bulky groups are present on same carbon atom of the double bond. Hence, isomerism is not possible. Therefore, the IUPAC name of the given molecule is 2-methyl-2-butene.
IUPAC name for the given molecule is assigned.
Want to see more full solutions like this?
Chapter 13 Solutions
General, Organic, and Biological Chemistry Seventh Edition
- Examine the structure shown below. A) What is the common name for this fatty acid? B) What is the IUPAC Name for this fatty acid? (Note: you can use the letter D to represent A in the name).arrow_forwardDraw the structure and write the IUPAC name of sertralinearrow_forwardPlease provide a chromatography technique to isolate the protein A from the mixture containing protein A and B (shown as below). And explain the separation mechanism related to this example. Protein A: MKRHRRKKHHRKRRKKRKGH (positively charged) Protein B: MDEEEEDDDEEDDEEDEDEED (negatively charged)arrow_forward
- Describe the principal biophysical and biochemical techniques used to study tertiary and quaternary structure of proteins.arrow_forwardThere are 4 classes of biochemical families (Carbohydrates, Proteins, Lipids, and Nucleic Acids). Identify the biochemical family that is described by the statements below. 1. The monomer of this biochemical family is called an amino acid. 2. DNA is an example of this biochemical family. 3. Glucose and sucrose are an example of this biochemical family. 4. Vitamin A, steroids, waxes, and phospholipids are all examples of this biochemical family. 5. This diverse biochemical family is known for being nonpolar and insoluble in water. 6. This biochemical family is the genetic material in an organism and is made up of a phosphate, sugar, and base monomer. 7. The monomer of this biochemical family contains an amine and a carboxylic acid functional group. Two monomers are linked together through condensation polymerization of these two functional groups.arrow_forwardName three biologically active peptides. Mention their functions.arrow_forward
- Using condensed structures, supply an outline of the mechanism for the formation of Gly-Gly in the human body. (Hint: It is not a simple condensation reaction).arrow_forwardResearch the IUPAC and common names and the short-hand codes of the following: 9 saturated fatty acids (4C-20C atoms) 5 unsaturated fatty acids (16C-20C atoms) 3 essential fatty acids 4 eicosanoidsarrow_forwardDraw the structure and name the following polypeptides: 1. SRAL b. ENIRYTAKarrow_forward
- Discuss one biologically active peptide. How important is it in biochemical processes?arrow_forwardWhen separated on a polyacrylamide gel, the procedure is abbreviated as SDS-PAGE (Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis). The technique is a standard means for separating proteins according to their molecular weight. The gels are neutral, hydrophilic, three-dimensional networks of long hydrocarbons cross-linked by methylene groups. (Give the main two compounds responsible for the formation of the gels)arrow_forwardWrite the structure of the following peptides: a) glycyl alanine b) tyrosyl alanyl glycine c) phenylalanyl tyrosyl seryl histidinearrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





