
Concept explainers
Interpretation:
The given reaction sequence has to be completed.
Concept Introduction:

Alkynes Synthesis:
Alkynes can be prepared from germinal dihalide or vicinal dihalide by treating with a very strong base.
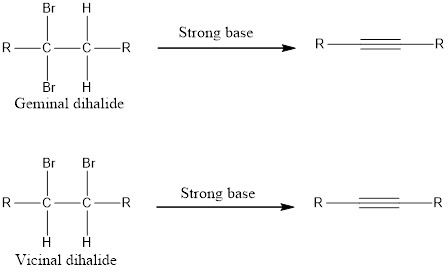
Two equivalents of base is required because the substrate is a dihalide. If the alkyne formed is a terminal alkyne, then three equivalents of base has to be used resulting in salt formation of the alkyne. This on treatment with water gives the terminal alkyne. The general reaction can be given as shown below,
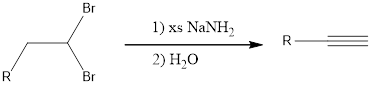
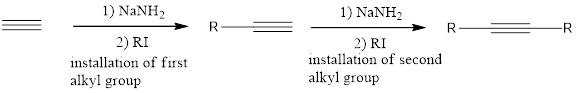
Substitution reaction of Alkynes:
The terminal alkynes can be deprotonated upon treatment with a strong base such as sodium amide to give an alkynide ion. Alkynide ions are very strong nucleophiles and can take part in

If the starting alkyne is acetylene, then the installation of the alkyl groups has to be done one at one time. This means that one group is installed on one side and then the second substitution takes place in next step.
Hydrogenation:
Hydrogenation reaction is one of the reactions that alkynes undergo. Addition of hydrogen is the hydrogenation reaction. This can be accomplished by using hydrogen molecule and metal catalyst. Hydrogenation reaction is also known as reduction reaction.
When an alkyne undergoes hydrogenation reaction with hydrogen and catalyst such as platinum, the final product obtained will be
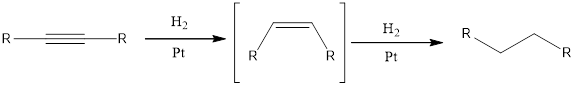
Under the above conditions, the alkene cannot be isolated. This is because the alkene is more reactive than the alkyne towards hydrogenation.
If the alkene has to be obtained from alkyne means, then a partially deactivated catalyst like Lindlar’s catalyst or sodium in liquid ammonia can be used. Partially deactivated catalyst is known as poisoned catalyst. When the alkyne is reduced using Lindlar’s catalyst, the alkene product obtained will be having cis configuration. If the alkyne is reduced using sodium in liquid ammonia means the alkene obtained will be having trans configuration.
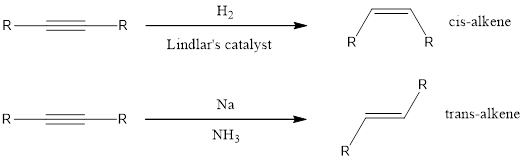
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Organic Chemistry As a Second Language: First Semester Topics
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





