
Concept explainers
Interpretation:
The value of
Concept introduction:
The wavefunction contains all the information about the state of the system. The wavefunction is the function of the coordinates of particles and time. The wavefunction
Answer to Problem 12.56E
The values of
The simple potential energy diagram for this system is shown in Figure 1 and Figure 2.
Explanation of Solution
The value of
Where,
•
•
The value of
Where,
•
The value of
The wavefunction
The value of
Substitute the value of
Thus, the value of
Substitute the value of
Thus, the value of
Substitute the value of
Thus, the value of
Substitute the value of
Thus, the wavefunction
The value of
Substitute the value of
Thus, the value of
Substitute the value of
Thus, the value of
Substitute the value of
Thus, the value of
Substitute the value of
Thus, the wavefunction
The value of
Substitute the value of
Thus, the value of
Substitute the value of
Thus, the value of
Substitute the value of
Thus, the value of
Substitute the value of
Thus, the wavefunction
The value of
Substitute the value of
Thus, the value of
Substitute the value of
Thus, the value of
Substitute the value of
Thus, the value of
Substitute the value of
Thus, the wavefunction
The values of potential energy and
| Energy |
|
The plot between the potential energy with
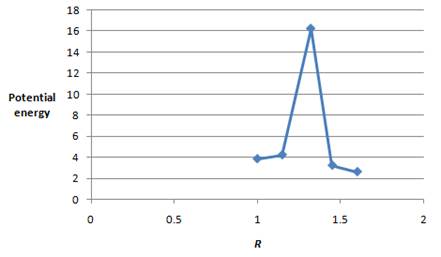
Figure 1
The values of potential energy and
| Energy |
|
The plot between the potential energy with
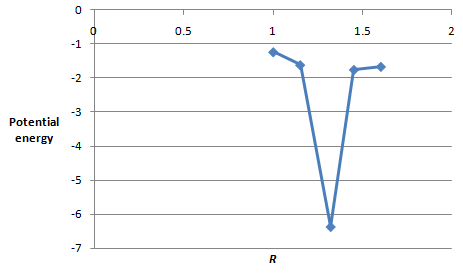
Figure 2
The values of
The simple potential energy diagram for this system is shown in Figure 1 and Figure 2.
Want to see more full solutions like this?
Chapter 12 Solutions
Physical Chemistry
- Decide how the sketches below would be listed, if they were listed in order of decreasing potential energy. That is, select "1" beside the sketch that shows the system with the highest potential energy, select "2" beside the sketch that shows the system with the next highest potential energy, and so on. Note for advanced students: since the units of each charge are not written, you may assume any convenient and reasonable unit, for example coulombs or multiples of e. +2 N +2 O 1 (highest) 2 3 4 (lowest) O 1 (highest) 2 3 4 (lowest) e +5 O 1 (highest) 02 0 3 ○ 4 (lowest) O 1 (highest) 2 3 O4 (lowest)arrow_forwardDecide how the sketches below would be listed, if they were listed in order of decreasing potential energy. That is, select "1" beside the sketch that shows the system with the highest potential energy, select "2" beside the sketch that shows the system with the next highest potential energy, and so on. Note for advanced students: since the units of each charge are not written, you may assume any convenient and reasonable unit, for example coulombs or multiples of e. +1 1 (highest) 1 (highest) alo 2 -1 +3 Ar 3 3 +3 4 (lowest) 4 (lowest) 1 (highest) 1 (highest) +1 2 +1 +5 3 3 -2 4 (lowest) 4 (lowest) Explanation Check © 2021 McGraw Hill LLC. AIl Rights Reserved. Terms of Use | Privacy Center | Accessibility O O O Oarrow_forwardDecide how the sketches below would be listed, if they were listed in order of decreasing potential energy. That is, select "1" beside the sketch that shows the system with the highest potential energy, select "2" beside the sketch that shows the system with the next highest potential energy, and so on. Note for advanced students: since the units of each charge are not written, you may assume any convenient and reasonable unit, for example coulombs or multiples of e. +1 +2 Explanation +3 -2 O 1 (highest) 02 03 O4 (lowest) 1 (highest) 02 3 O4 (lowest) Check 80 +3 +5 +1 F4 X 29 O 1 (highest) 02 03 O4 (lowest) O 1 (highest) 02 W N F5 3 O4 (lowest) S M F6 tv ♫ © 2023 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility O all A Sh 9 A CHO F7 FB ol А Ar PAarrow_forward
- What is the electrostatic potential energy between an electron and a proton that are separated by 53 pm?arrow_forward2. Calculate the enthalpy change for the reaction : C4H4 (9) + H2 (9) C2H6 (g) Given the following data: i) C2H4 (9) + 3 O2 (9) → 2 CO2 (9) + 2 H2O (1) AH° = - 1410.9 kJ 2 C2H6 (9) + 7 02 (9) →4 CO2 (9) + 6 H20 (1) ii) iii) 2 H2 (9) + O2 (g) 2 H2O (1) AH° = - 3119.4 kJ AH° = - 571.6 kJ 3. Calculate the standard enthalpy change for the reaction, C2H2 (g) + 2 H2 (9g) - C2H6 (9) Given that: i) 2 C2H2 (9) + 5 02 (g) 4 CO2 (9) + 2 H2O (1) AH° = - 2599 kJ 3119 kJ ii) 2 C2H6 (9) + 7 02 (9) - 4 CO2 (9) + 6 H20 (1) AH° = iii) 2 H2 (9) + O2 (9) → 2 H2O () AH° = - 572 kJ The enthalpy of combustion of methane is -891 kJmol-1. Calculate the enthalpy of formation of methane if the enthalpy of formation of carbon dioxide and water are - 394 kJmol-1 and -286 kJmol respectively. Chemical equation for formation of 4. methane: C(s) + 2H2(9) CH4(9) Given: AH = -891 kJmol-1 CH4(9) + 202(9) 2H2O() + CO2(g) C(s) + Oz(g) → CO2(9) H2(g) + 2 O2 - H20 AH = -394 kJmol-1 AH = -286 kJmol-1arrow_forwardDecide how the sketches below would be listed, if they were listed in order of decreasing potential energy. That is, select "1" beside the sketch that shows the system with the highest potential energy, select "2" beside the sketch that shows the system with the next highest potential energy, and so on. Note for advanced students: since the units of each charge are not written, you may assume any convenient and reasonable unit, for example coulombs or multiples of e. I Don't Know -1 +4 +2 Submit . . O 1 (highest) 2 03 O4 (lowest) O 1 (highest) 02 03 O4 (lowest) +1 +1 +1 +1 MA Xx O 1 (highest) 02 234 4 (lowest) O 1 (highest) 02 O 3 O4 (lowest) €23 ~ tv 0 Zoom ? Ⓒ2023 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Access 9 NZA Pud ol Aarrow_forward
- Use the molar bond enthalpy data in the table to estimate the value of AHxn for the equation NH, (g) + 20,(g) → HNO, (g) + H,O(g) The bonding in the molecules is shown. + H + O=O H.arrow_forwardDecide how the sketches below would be listed, if they were listed in order of decreasing potential energy. That is, select "1" beside the sketch that shows the system with the highest potential energy, select "2" beside the sketch that shows the system with the next highest potential energy, and so on. Note for advanced students: since the units of each charge are not written, you may assume any convenient and reasonable unit, for example coulombs or multiples of e. ✪ O 1 (highest) 02 03 O 4 (lowest) O 1 (highest) 02 03 O 4 (lowest) @ +2 O 1 (highest) 02 0 3 O4 (lowest) O 1 (highest) 02 03 O 4 (lowest) $arrow_forward3. Calculate the reaction enthalpy of this reaclion Under Stondard conditions. 2C, Hu (G) + 70, lg) ->4C0, 4) + (6 H,O ) 70, (g) –> 4CO,(G) +(6 HqO() kJarrow_forward
- Construct the expression for Kc for the following reaction. C(s) + H,O(g) = CO(g) + H,(g)arrow_forwarda) The value for Ψ in root tissue was found to be -3.3 bars. If you take the root tissue and place it in a 0.1 M solution of sucrose at 20°C in an open beaker, what is the Ψ of the solution, and in which direction would the net flow of water be? b) NaCl dissociates into 2 particles in water: Na+ and Cl-. If the solution in question 4 contained 0.1M NaCl instead of 0.1M sucrose, what is the Ψ of the solution, and in which direction would the net flow of water be? c) A plant cell with a Ψs of -7.5 bars keeps a constant volume when immersed in an open-beaker solution that has a Ψs of -4 bars. What is the cell’s ΨP?arrow_forwardDetermine E, H, G, and S for CH4 at standard pressure and 25°C. σ equals 12 for methane and the atomization energy of CH4 is 1163 kJ/mol.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
