
(a)
Interpretation:
The appropriate alkene or alkyne from which the given compound can be produced is to be determined along with the necessary reagents and special reaction conditions.
Concept introduction:
Oxidation of
The initial addition of borane is a syn, anti-Markovnikov addition via a four-membered cyclic transition state. Boron adds to the less substituted (less hindered) carbon because it is less electronegative than hydrogen. This results in an anti-Markovnikov addition of a water molecule to the double bond.
Answer to Problem 12.53P
The appropriate alkene used to synthesize the given compound is
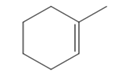
The necessary reagents and special reaction conditions for the synthesis are
Explanation of Solution
The structure of the given compound is
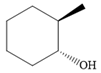
Alcohol with a specific stereochemistry is to be synthesized from an alkene. A reaction involving a carbocation needs to be avoided to prevent unwanted carbocation rearrangements. Also, the OH group must be added to a less substituted carbon, i.e., an anti-Markovnikov addition is needed. Therefore, the reaction needs to be carried out using hydroboration-oxidation.
The appropriate alkene for the synthesis of the given compound would be
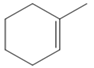
The given compound is synthesized by using the above alkene via hydroboration-oxidation reaction. So the necessary reagents for the reaction are
In the first step, an electrophilic addition of borane across the double bond of the alkene takes place either from above or below the plane of the alkene. So a mixture of enantiomers is obtained after oxidation of the adduct by basic
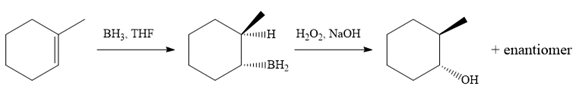
The alkene necessary for the synthesis of the given compound and the reagents and special conditions for the reaction are determined on the basis of the structure of the given compound.
(b)
Interpretation:
The appropriate alkene or alkyne from which the given compound can be produced is to be determined along with the necessary reagents and special reaction conditions.
Concept introduction:
Oxidation of alkenes and alkynes is carried out by the hydroboration-oxidation reaction. Alkenes are oxidized to alcohol while alkynes are oxidized to the corresponding carbonyl compounds. Terminal alkynes are oxidized to the corresponding aldehyde, and internal alkynes are oxidized to the corresponding ketone by a sterically hindered borane like disiamylborane
The initial addition of borane is a syn, anti-Markovnikov addition via a four-membered cyclic transition state. Boron adds to the less substituted (less hindered) carbon because it is less electronegative than hydrogen. This results in an anti-Markovnikov addition of a water molecule to the double bond.
Answer to Problem 12.53P
The appropriate alkyne used to synthesize the given compound is
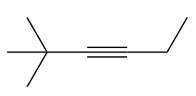
The necessary reagents and special reaction conditions to synthesize the given compound are
Explanation of Solution
The given compound is
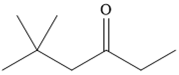
It is a ketone, so the starting compound must be an alkyne. A hydroboration-oxidation reaction can convert an alkyne into a ketone. Since only one molecule of borane is to be added, a bulky reagent like disiamylborane is more appropriate than borane. Also, the dialkylborane part must add to the less hindered carbon of the triple bond. Therefore, the triple bond must be between the carbon bonded to oxygen and the carbon close to the bulky tertiary carbon. Therefore, the alkyne that can be used is

A sterically hindered dialkylborane, like disiamylborane
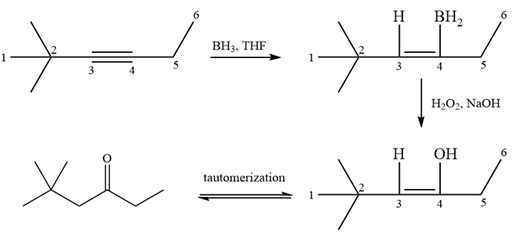
Thus the specific reagents and reaction conditions are
The alkene necessary for the synthesis of the given compound and the reagents and special conditions for the reaction are determined on the basis of the structure of the given compound.
(c)
Interpretation:
The appropriate alkene or alkyne from which the given compound can be produced is to be determined along with the necessary reagents and special reaction conditions.
Concept introduction:
Oxidation of alkenes and alkynes is carried out by the hydroboration-oxidation reaction. Alkenes are oxidized to alcohol while alkynes are oxidized to the corresponding carbonyl compounds. Terminal alkynes are oxidized to the corresponding aldehyde, and internal alkynes are oxidized to the corresponding ketone by a sterically hindered borane like disiamylborane
The initial addition of borane is a syn, anti-Markovnikov addition via a four-membered cyclic transition state. Boron adds to the less substituted (less hindered) carbon because it is less electronegative than hydrogen. This results in an anti-Markovnikov addition of a water molecule to the double bond.
Answer to Problem 12.53P
The appropriate alkyne used to synthesize the given compound is
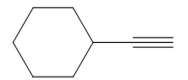
The necessary reagents and special reaction conditions to synthesize the given compound are
Explanation of Solution
The given compound is
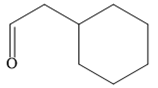
It is an aldehyde, so it can be prepared from a terminal alkyne by hydroboration-oxidation. In the hydroboration reaction, boron is added to the terminal carbon. So the appropriate alkyne for the synthesis of the given compound is
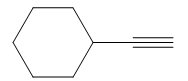
The alkyne is treated with the bulky disiamylborane to prevent the addition of a second molecule and formation of a mixture of products. Subsequent treatment of the adduct by
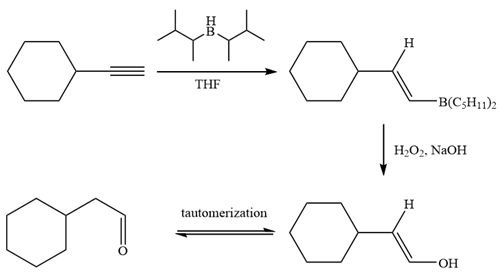
Thus, the necessary reagents and special reaction conditions for the synthesis are
The alkene necessary for the synthesis of the given compound and the reagents and special conditions for the reaction are determined on the basis of the structure of the given compound.
(d)
Interpretation:
The appropriate alkene or alkyne from which the given compound can be produced is to be determined along with the necessary reagents and special reaction conditions.
Concept introduction:
Oxidation of alkenes and alkynes is carried out by the hydroboration-oxidation reaction. Alkenes are oxidized to alcohol while alkynes are oxidized to the corresponding carbonyl compounds. Terminal alkynes are oxidized to the corresponding aldehyde, and internal alkynes are oxidized to the corresponding ketone by a sterically hindered borane like disiamylborane
The initial addition of borane is a syn, anti-Markovnikov addition via a four-membered cyclic transition state. Boron adds to the less substituted (less hindered) carbon because it is less electronegative than hydrogen. This results in an anti-Markovnikov addition of a water molecule to the double bond.
Answer to Problem 12.53P
The appropriate alkene used to synthesize the given compound is
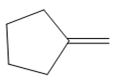
The necessary reagents and special reaction conditions for the synthesis are
Explanation of Solution
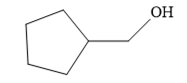
Since the product is an alcohol, an alkene with a methylene substituent on a cyclopentane ring would be appropriate as the starting compound.

Treating this alkene with borane in THF will add
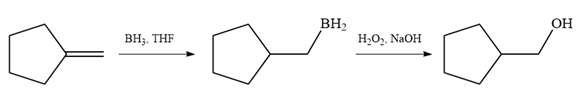
Thus, the necessary reagents for the reaction are
The alkene necessary for the synthesis of the given compound and the reagents and special conditions for the reaction are determined on the basis of the structure of the given compound.
Want to see more full solutions like this?
Chapter 12 Solutions
Organic Chemistry: Principles And Mechanisms
- (12) Which one of the following statements about fluo- rometry is FALSE? a) Fluorescence is better detected at 90 from the exci- tation direction. b) Fluorescence is typically shifted to longer wave- length from the excitation wavelength. c) For most fluorescent compounds, radiation is pro- duced by a transitionarrow_forwardDon't used Ai solutionarrow_forwardDon't used Ai solutionarrow_forward
- Don't used Ai solutionarrow_forwardIndicate the correct option.a) Graphite conducts electricity, being an isotropic materialb) Graphite is not a conductor of electricityc) Both are falsearrow_forward(f) SO: Best Lewis Structure 3 e group geometry:_ shape/molecular geometry:, (g) CF2CF2 Best Lewis Structure polarity: e group arrangement:_ shape/molecular geometry: (h) (NH4)2SO4 Best Lewis Structure polarity: e group arrangement: shape/molecular geometry: polarity: Sketch (with angles): Sketch (with angles): Sketch (with angles):arrow_forward
- 1. Problem Set 3b Chem 141 For each of the following compounds draw the BEST Lewis Structure then sketch the molecule (showing bond angles). Identify (i) electron group geometry (ii) shape around EACH central atom (iii) whether the molecule is polar or non-polar (iv) (a) SeF4 Best Lewis Structure e group arrangement:_ shape/molecular geometry: polarity: (b) AsOBr3 Best Lewis Structure e group arrangement:_ shape/molecular geometry: polarity: Sketch (with angles): Sketch (with angles):arrow_forward(c) SOCI Best Lewis Structure 2 e group arrangement: shape/molecular geometry:_ (d) PCls Best Lewis Structure polarity: e group geometry:_ shape/molecular geometry:_ (e) Ba(BrO2): Best Lewis Structure polarity: e group arrangement: shape/molecular geometry: polarity: Sketch (with angles): Sketch (with angles): Sketch (with angles):arrow_forwardDon't used Ai solutionarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





