
(a)
Interpretation:
Considering the acid catalyzed mechanism of ester hydrolysis reaction answer the following,
The species which can be represented by
Concept Introduction:
An acid catalyzed hydrolysis of ester is much faster reaction as compared to uncatalyzed hydrolysis of ester. The acid catalyzed reaction mechanism is written as,
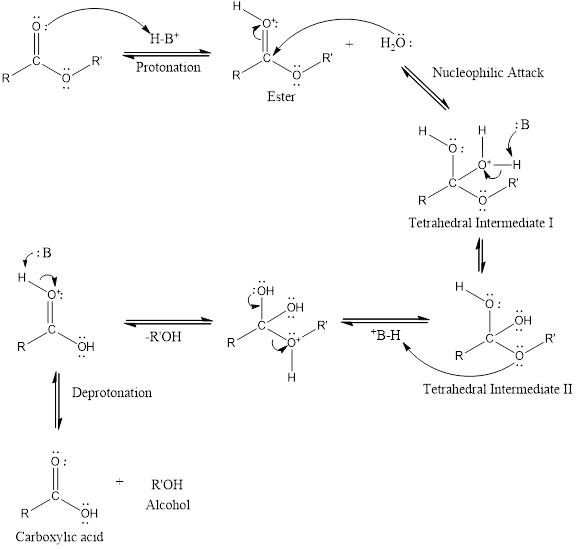
(b)
Interpretation:
Considering the acid catalyzed mechanism of ester hydrolysis reaction answer the following,
The species which can be represented by
Concept Introduction:
An acid catalyzed hydrolysis of ester is much faster reaction as compared to uncatalyzed hydrolysis of ester. The acid catalyzed reaction mechanism is written as,
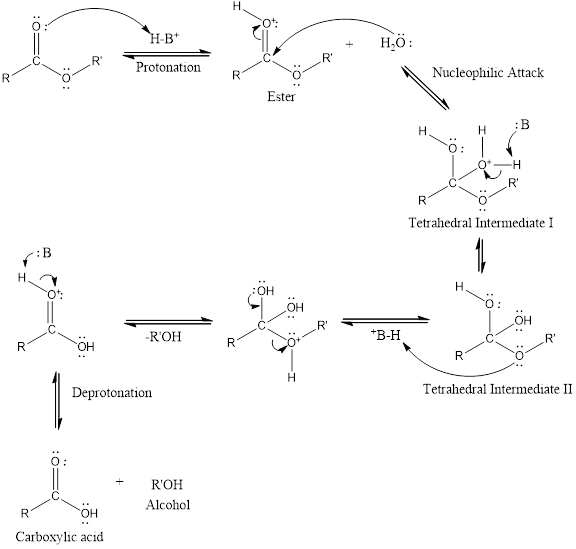
(c)
Interpretation:
Considering the acid catalyzed mechanism of ester hydrolysis reaction answer the following,
The species which is mostly used as
Concept introduction:
An acid catalyzed hydrolysis of ester is much faster reaction as compared to uncatalyzed hydrolysis of ester. The acid catalyzed reaction mechanism is written as,

(d)
Interpretation:
Considering the acid catalyzed mechanism of ester hydrolysis reaction answer the following,
The species which is mostly used as
Concept Introduction:
An acid catalyzed hydrolysis of ester is much faster reaction as compared to uncatalyzed hydrolysis of ester. The acid catalyzed reaction mechanism is written as,

Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
EBK ESSENTIAL ORGANIC CHEMISTRY
- Which of the following molecules is most nucleophilic? OA. NaOCH₂CH3 OB. CH3CH₂OH OC. NaOC6H5 OD. C₂H5OH 1 E.CH3COONaarrow_forwardHi, For this proton transfer, why does the NH3 electron pair move to the H and not the electron pair from the Br?arrow_forwardWhich one of the following synthetic routes gives the best yield of meta-bromoaniline starting with benzene? NH2 HNO, 1) Sn. HCI Br, A. FeBr, H,SO, 2) NaOH Br metabromoaniline 1) Sn, HCI Br, HNO, В. H,SO, 2) NaOH FeBr, HNO, Br, 1) Sn, HCI C. FeBr H,SO, 2) NaOH 1) Sn, HCI HNO Br, D. 2) NaOH HSO FeBr, O A O B O C Ouestion 14 6 pts MacBook Pro Search or type URLarrow_forward
- The reaction of a nitrile with an alcohol in the presence of a strong acid forms an N-substituted amide. This reaction, known as the Ritter reaction, doesnot work with primary alcohols. a. Why does the Ritter reaction not work with primary alcohols? b. Provide an explanation for why an amide is less susceptible to nucleophilic attack than its corresponding ester.arrow_forwardIn the following reaction which species acts as the nucleophile? OCH CH3 (H.C).NOCH CH CH,CH OH OCH,CH , OCH,CH, A. OCH,CH, (H,C),N B. C. CH,CH,OH D. (D)arrow_forwardA. Rank the compounds (shown below) in order of increasing acidity. ов A B C E B: Choose the reagent(s) that would be most likely to complete this reaction. ||| > | > | ||| | > || D ||| | > ||arrow_forward
- For each of the reactions indicate which reactant is the nucleophile and which is the electrophile.arrow_forwardZ. Whch one of the following reagents will not transform an aldehyde to a carboxylic acid? с) Месоон E) HOA9(NH3)2 then H30* D) Zn(Hg), HCI A) CrOs, HSO4 B) KMNO4, H2S04 8. Which one of the following reactions will not produce phenol (PHOH)? PHOCH3 PHOCCH, PhCCHs PhMgBr PhN2* 1) MečoOH H2O Cu,O, H2O 1) LIAIH4 HBr, heat 2) H20, H;O*, heat 2) H2O, H3O* C 9. Which one of the following compounds will not react with F3CC=CCF3 in a Diels-Alder reaction OMe B C 10. Which one of the following compounds is anti-aromatic? HN: : NH A B C 4arrow_forwardIdentify the products obtained by the hydrolysis of the given compound. оо | | -COC- Select one: O a. one molecule of dibenzoic acid and one molecule of benzene O b. one molecule of benzene and one molecule of a carboxylic acid O c. two molecules of phenol O d. two molecules of benzoic acidarrow_forward
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

