
(a)
Interpretation:
The acyl chloride and carboxylate ion that could be used to form the given mixed anhydride have to be found.
Concept Introduction:
Mixed anhydrides are unsymmetrical anhydrides which have different alkyl groups.
Example:
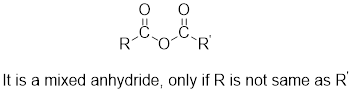
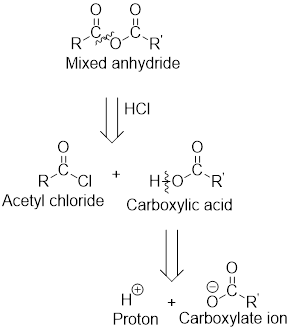
The acetyl chloride and carboxylate ion that could be used to form any given mixed anhydride can be analysed using the simple retro analysis as shown here:
(b)
Interpretation:
The other reagents that could be used to form the given mixed anhydride have to be found.
Concept Introduction:
Mixed anhydrides are unsymmetrical anhydrides which have different alkyl groups.
Example:
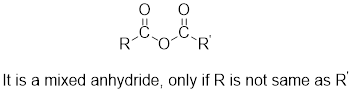
Any anhydride can be obtained using the following two pairs of reagents:
- 1. Acetyl chloride and a carboxylate ion.
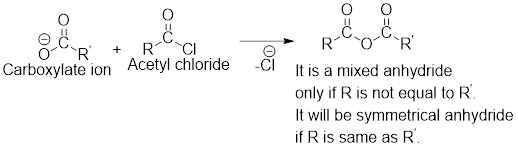
- 2. Two different carboxylic acids if mixed or unsymmetrical anhydride has to be formed or two same carboxylic acids if symmetrical anhydride has to be formed.
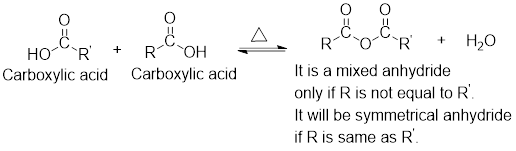
For any simple anhydride, the two carboxylic acids from which it has been formed can be found by the simple retro analysis as shown here:
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
EBK ESSENTIAL ORGANIC CHEMISTRY
- 1) Answer the following questions: a. Explain oxidative and reductive Ozonolysis. b. Give the sequence of the relative reactivities of carboxylic acid derivatives. Explain why acyl chloride can be converted into ester but ester cannot be converted into acyl chloride? c. Write the product when ester of propionic acid, propionyl chloride and propanal reacts with excess of methyl magnesium bromidearrow_forwardWhich of these statements is NOT true about the reaction of amines with a cyclic anhydride like phthalic anhydride? a. Dimethylamine will give a cyclic imide product. b. Aniline will give a cyclic imide product. c. Triethylamine will give a cyclic imide product. d. The reaction undergoes a nucleophilic acyl substitution mechanism.arrow_forwardExplain why acetyl chloride reacts faster with water than acetic anhydride does?arrow_forward
- 1. Give the product of the reaction of the cyclic anhydride shown below when treated with the following reagents. a. aquous NaOH b. aqueous HCI c. methanol d. sodium methoxide in methanol e. ethylaminearrow_forward1. Put these three common types of carbonyl compound in order of decreasing reactivity ester amide acid chloride 2. For the least reactive, show the interconversion to its other resonance form: How does this electron delocalisation make it stable? 3. For the most reactive, draw the mechanism of its undergoing hydrolysis (reaction with H2O): Why makes this type of carbonyl so reactive to nucleophiles?arrow_forwardPlease explain the chosen letter. Benzoic acid reacts with conc. HNO3 and conc.H2SO, to give a. o-nitrobenzoic acid b. p-nitrobenzoic acid c. m-nitrobenzoic acid d. p-dinitrobenzoic acidarrow_forward
- Please answer each part A B and Carrow_forwardDraw the structure of the product of the Michael reaction between propenamide and diethyl malonatearrow_forward1. Occasionally, a crossed Cannizzaro reaction is carried out to reduce benzaldehyde derivatives to the corresponding benzyl alcohol, utilizing formaldehyde as the reducing agent. i) Please show the mechanism by which this occurs.arrow_forward
- What reagents would you use to convert methyl propanoate to the following compounds? a. isopropyl propanoate b. sodium propanoate c. N-ethylpropanamide d. propanoic acidarrow_forwardWhat is the first step in the general mechanism for electrophilic aromatic substitution? a. aromatic ring protonation b.loss of the electrophilic aromatic ring c. deprotonation of the aromatic ring d.addition of the electrophilic to the aromatic ring.arrow_forwardImagine if one replaced acetic anhydride with benzoic anhydride (shown). Draw the structure of the new product of the reaction with salicylic acid.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


