
(a)
Interpretation:
To predict the product of acid-catalyzed hydrolysis of given ester.
Concept Introduction:
An acid-catalyzed hydrolysis of the ester is a much faster reaction as compared to uncatalyzed hydrolysis of the ester. The addition of acid promotes the protonation of oxygen atom in the carbonyl group and as it is a fact that an oxygen atom with positive charge has more electron withdrawing tendency than neutral atom. The more withdrawal of electron density of oxygen atom decreases the electron density from the carbonyl carbon and make it more susceptible for the attack of the nucleophile. The acid catalyzed reaction mechanism is written as,
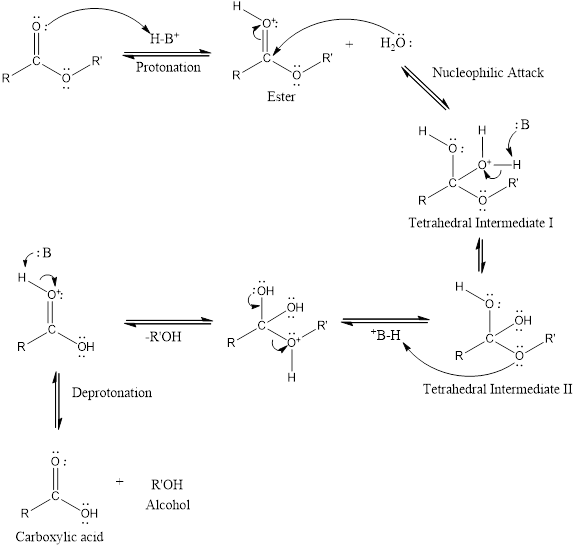
Therefore, products obtained by the acid catalyzed ester hydrolysis are the
(b)
Interpretation:
To predict the product of acid-catalyzed hydrolysis of given ester.
Concept introduction:
An acid-catalyzed hydrolysis of the ester is a much faster reaction as compared to uncatalyzed hydrolysis of the ester. The addition of acid promotes the protonation of oxygen atom in the carbonyl group and as it is a fact that an oxygen atom with positive charge has more electron withdrawing tendency than neutral atom. The more withdrawal of electron density of oxygen atom decreases the electron density from the carbonyl carbon and make it more susceptible for the attack of the nucleophile. The acid catalyzed reaction mechanism is written as,
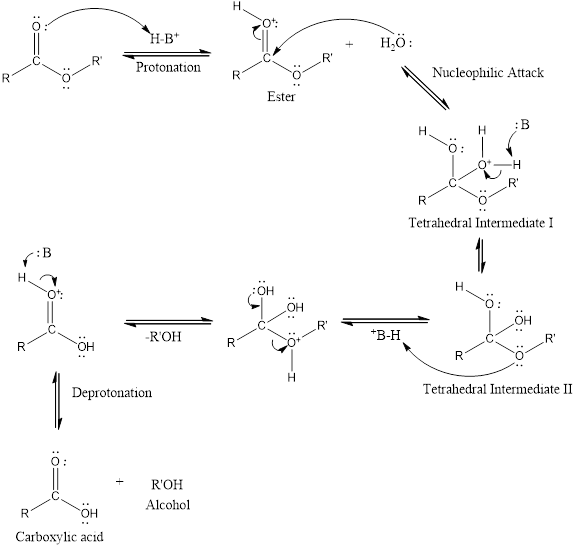
Therefore, products obtained by the acid catalyzed ester hydrolysis are the carboxylic acid and an alcohol by which ester was formed.
(c)
Interpretation:
To predict the product of acid-catalyzed hydrolysis of given ester.
Concept Introduction:
An acid-catalyzed hydrolysis of the ester is a much faster reaction as compared to uncatalyzed hydrolysis of the ester. The addition of acid promotes the protonation of oxygen atom in the carbonyl group and as it is a fact that an oxygen atom with positive charge has more electron withdrawing tendency than neutral atom. The more withdrawal of electron density of oxygen atom decreases the electron density from the carbonyl carbon and make it more susceptible for the attack of the nucleophile. The acid catalyzed reaction mechanism is written as,

Therefore, products obtained by the acid catalyzed ester hydrolysis are the carboxylic acid and an alcohol by which ester was formed.
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Essential Organic Chemistry (3rd Edition)
- What is the major advantage of base-catalyzed hydrolysis of esters over acid-catalyzed hydrolysis?arrow_forwardDraw the two products that form when this ester is treated with H2O and H2SO4. Oarrow_forwardIn which condition can carboxylic acids undergo nucleophilic acyl substitution reactions?arrow_forward
- Draw the structures of the products from the hydrolysis of each of the following with NaOHNaOH.arrow_forwardDraw the structures of the products from the hydrolysis of each of the following with NaOHNaOH.arrow_forwardIn biochemical reactions, decarboxylation of carboxylic acids typically takes place for-keto carboxylic acids. Justify a rational why nature opted for-keto carboxylic acid decarboxylation. Among the following types of biochemical reactions, ester hydrolysis, rearrangement reactions, water elimination reactions, and anhydride hydrolyses, which one is the most favorable one. Rank the above reactions types in the order of being the most to least favorable reactionarrow_forward
- Predict the products formed when cyclohexanone reacts with the following reagents.phenylhydrazine and weak acidarrow_forwardQ: What ester results in the following reflux? pentanoic acid CH₂CH₂OH OH R' R-OH Q: What alcohol and acid must we use to make the following ester? O O H+ OH H+ benzyl isobutanoate H₂Oarrow_forwardOne frequently used method for preparing methyl esters is by reaction of carboxylic acids with diazomethane, CH2N2. The reaction occurs in two steps: (l) protonation of diazomethane by the carboxylic acid to yield methyldiazonium ion, CH3N2+, plus a carboxylate ion; and (2) reaction of the carboxylate ion with CH3N2+. (a) Draw two resonance structures of diazomethane, and account for step 1. (b) What kind of reaction occurs in step 2?arrow_forward
- I am very confused and I want to study so please help me with these problemsarrow_forwardWhat products are formed by acidic hydrolysis of the following compound? benzaldehyde and butan-1-d benzeie acid and butan-2-d 2-methylbutanoic acid and phenol benzoic acid and methyl ethyl ketonearrow_forwardExplain why carboxylic acids do not undergo nucleophilic acyl substitution reactions with amines.arrow_forward

 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning





