
Essential Organic Chemistry (3rd Edition)
3rd Edition
ISBN: 9780321937711
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Question
Chapter 11.15, Problem 29P
(a)
Interpretation Introduction
Interpretation: The acyl chloride and amine that would be required to synthesize the given amide have to be found.
Concept Introduction:
The reactions of acetyl chloride with
On reaction with amines, acetyl chloride forms amides.
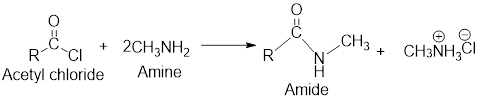
The retro analysis for finding the starting materials of the given amide is shown here:
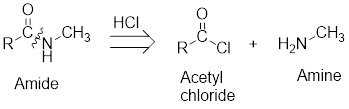
(b)
Interpretation Introduction
Interpretation: The acyl chloride and amine that would be required to synthesize the given amide have to be found.
Concept Introduction:
The reactions of acetyl chloride with amines:
On reaction with amines, acetyl chloride forms amides.

The retro analysis for finding the starting materials of the given amide is shown here:
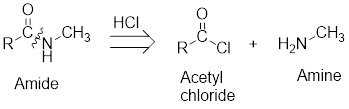
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
From the given structures which is(a) amide that will release a secondary amine upon hydrolysis? (b) product of hydrolysis of MSO (c) a tertiary amide and (d) a diketone
N-p-hydroxyphenylethanamide is commonly known as
a. acetaminophen
b. acetamide
c. acetanilide
d. formamide
High molar mass amines have __________ odor.
a.strong ammoniacal
b.fruity
c.fishy
d.obnoxious
Trimethyl amine has _________ odor.
a.obnoxious
b.fishy
c. ammoniacal
d. fruity
Aromatic amines are _________
neutral because of the polarity
are unique as they do not behave like any of the other amines
acidic, but stronger than the aliphatic amines
basic, but weaker than aliphatic amines
Chapter 11 Solutions
Essential Organic Chemistry (3rd Edition)
Ch. 11.1 - The aromas of many flowers and fruits are due to...Ch. 11.1 - Name the following compounds:Ch. 11.1 - Prob. 3PCh. 11.2 - Prob. 4PCh. 11.2 - Prob. 5PCh. 11.4 - a. What is the product of the reaction of acetyl...Ch. 11.4 - Prob. 7PCh. 11.5 - Using the pKa values listed in Table 11.1, predict...Ch. 11.6 - Starting with acetyl chloride, what neutral...Ch. 11.6 - Prob. 10P
Ch. 11.7 - Prob. 11PCh. 11.8 - Prob. 13PCh. 11.8 - Using the mechanism for the acidcatalyzed...Ch. 11.8 - Prob. 15PCh. 11.8 - Prob. 16PCh. 11.8 - Prob. 17PCh. 11.9 - Prob. 18PCh. 11.10 - Show how each of the following esters could be...Ch. 11.11 - Which of the following reactions would lead to the...Ch. 11.12 - Prob. 22PCh. 11.12 - Prob. 23PCh. 11.13 - Prob. 24PCh. 11.13 - Prob. 25PCh. 11.14 - Prob. 26PCh. 11.14 - Prob. 27PCh. 11.14 - Prob. 28PCh. 11.15 - Prob. 29PCh. 11.15 - How would you synthesize the following compounds...Ch. 11 - Write a structure for each of the following a. N,N...Ch. 11 - Prob. 32PCh. 11 - Which ester is more reactive, methyl acetate or...Ch. 11 - What products would be formed from the reaction of...Ch. 11 - What products would be obtained from the following...Ch. 11 - Prob. 36PCh. 11 - a. Which compound would you expect to have a...Ch. 11 - a. List the following esters in order of...Ch. 11 - D. N. Kursanov, a Russian chemist, proved that the...Ch. 11 - Prob. 40PCh. 11 - Using an alcohol for one method and an alkyl...Ch. 11 - Prob. 42PCh. 11 - Prob. 44PCh. 11 - Prob. 45PCh. 11 - Prob. 46PCh. 11 - Prob. 47PCh. 11 - Prob. 48PCh. 11 - Prob. 49PCh. 11 - Show how the following compounds could be prepared...Ch. 11 - Prob. 51PCh. 11 - Prob. 52PCh. 11 - Prob. 53P
Knowledge Booster
Similar questions
- Acetaminophen is an analgesic marketed under the brand name Tylenol, among others. Draw the amine that results from the base hydrolysis of acetaminophen.arrow_forwardWhich type of amine is phentermine? a) a primary aliphatic amine b) a primary aromatic amine c) a tertiary aliphatic amine d) a tertiary aromatic aminearrow_forwardDraw the structure of the product that will be formed when each of the following amines reacts with sodium nitrite and hydrochloric acid, followed by cuprous chloride. (a) propylamine (b) dipropylamine (c) N-propylaniline (d) N,N-dipropylaniline (e) p-propylanilinearrow_forward
- Which type of amine is (s)-methamphetamine? a) a primary aliphatic amine b) a primary aromatic amine c) a secondary aliphatic amine d) a secondary aromatic aminearrow_forwarda. Explain the effect of acid on the solubility of the water-insoluble amines.arrow_forwardReactions of aldehydes and ketones with amines and amine derivatives a. Draw reaction with a primary amine forms an imine. Hydrazine and hydroxylamine can also be used; they form a hydrazone and an oxime, respectively. b. Draw reaction with a secondary amine forms an enamine.arrow_forward
- Draw a structural formula for each amine and amine derivative. (a) N,N-Dimethylaniline (b) Triethylamine (c) tert-Butylamine (d) 1,4-Benzenediamine (e) 4-Aminobutanoic acid (f) (R)-2-Butanamine (g) Benzylamine (h) trans-2-Aminocyclohexanol (i) 1-Phenyl-2-propanamine (amphetamine) (j) Lithium diisopropylamide (LDA) (k) Benzyltrimethylammonium hydroxide (Triton B)arrow_forwardIndicate whether the following statement is true or false. Aliphatic amines are more basic than ammonia, whereas aromatic amines are less basic than ammonia.RightFalsearrow_forwardReductive amination of carbonyl compounds produces the same kinds of products as does the reaction of amines and alkyl halides True or Falsearrow_forward
- Draw the structure of the product that will be formed when each of the following amines reacts with sodium nitrite and hydrochloric acid, followed by cuprous chloride. (e) p-propylanilinearrow_forwardThe presence of amides in living organisms is beneficial due its stability which results from being the least reactive carboxylic acid derivative. True or Falsearrow_forwarda. What pH would you make the water layer to cause the carboxylic acid to dissolve in the water layer and the amine to dissolve in the ether layer? b. What pH would you make the water layer to cause the carboxylic acid to dissolve in the ether layer and the amine to dissolve in the water layer?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning