
Consider a two-stage compression refrigeration system operating between the pressure limits of 1.4 and 0.12 MPa. The working fluid is refrigerant-134a. The refrigerant leaves the condenser as a saturated liquid and is throttled to a flash chamber operating at 0.5 MPa. Part of the refrigerant evaporates during this flashing process, and this vapor is mixed with the refrigerant leaving the low-pressure compressor. The mixture is then compressed to the condenser pressure by the high-pressure compressor. The liquid in the flash chamber is throttled to the evaporator pressure, and it cools the refrigerated space as it vaporizes in the evaporator. Assuming the refrigerant leaves the evaporator as saturated vapor and both compressors are isentropic, determine (a) the fraction of the refrigerant that evaporates as it is throttled to the flash chamber, (b) the amount of heat removed from the refrigerated space and the compressor work per unit mass of refrigerant flowing through the condenser, and (c) the coefficient of performance.
(a)
The fraction of the refrigerant that evaporates as it is throttled to the flash chamber.
Answer to Problem 113RP
The fraction of the refrigerant that evaporates as it is throttled to the flash chamber is
Explanation of Solution
Show the T-s diagram as in Figure (1).
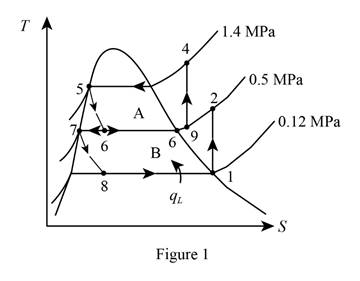
From Figure (1), write the specific enthalpy at state 5 is equal to state 6 due to throttling process.
Here, specific enthalpy at state 5 and 6 is
From Figure (1), write the specific enthalpy at state 7 is equal to state 8 due to throttling process.
Here, specific enthalpy at state 7 and 8 is
Express the fraction of the refrigerant that evaporates as it is throttled to the flash chamber
Here, specific enthalpy at saturated vapor is
Conclusion:
Perform unit conversion of pressure at state 1 from
Refer Table A-12, “saturated refrigerant-134a-pressure table”, and write the properties corresponding to pressure at state 1
Here, specific entropy and enthalpy at state 1 is
Refer Table A-13, “superheated refrigerant 134a”, and write the specific enthalpy at state 2 corresponding to pressure at state 2 of
Write the formula of interpolation method of two variables.
Here, the variables denote by x and y is specific entropy at state 2 and specific enthalpy at state 2 respectively.
Show the specific enthalpy at state 2 corresponding to specific entropy as in Table (1).
|
Specific entropy at state 2 |
Specific enthalpy at state 2 |
| 0.9384 | 263.48 |
| 0.94789 | |
| 0.9704 | 273.03 |
Substitute
Thus, the specific enthalpy at state 2 is,
Perform unit conversion of pressure at state 3 from
Refer Table A-12, “saturated refrigerant-134a-pressure table”, and write the property corresponding to pressure at state 3
Perform unit conversion of pressure at state 5 from
Refer Table A-12, “saturated refrigerant-134a-pressure table”, and write the property corresponding to pressure at state 5
Here, specific enthalpy at saturated liquid is
Substitute
Refer Table A-12, “saturated refrigerant-134a-pressure table”, and write the property corresponding to pressure at state 8
Substitute
Refer Table A-12, “saturated refrigerant-134a-pressure table”, and write the specific enthalpy at evaporation and pressure of
Substitute
Hence, the fraction of the refrigerant that evaporates as it is throttled to the flash chamber is
(b)
The amount of heat removed from the refrigerated space and the compressor work per unit mass of refrigerant flowing through the condenser.
Answer to Problem 113RP
The amount of heat removed from the refrigerated space is
Explanation of Solution
Express the enthalpy at state 9 by using an energy balance on the mixing chamber.
Here, the rate of total energy entering the system is
Express the amount of heat removed from the refrigerated space.
Express the compressor work per unit mass of refrigerant flowing through the condenser.
Conclusion:
Substitute
Refer Table A-13, “superheated refrigerant 134a”, and write the specific entropy at state 9 corresponding to pressure at state 9 of
Show the specific enthropy at state 9 corresponding to specific enthalpy as in Table (2).
|
Specific enthalpy at state 9 |
Specific entropy at state 9 |
| 263.48 | 0.9384 |
| 264.28 | |
| 273.03 | 0.9704 |
Use excels and tabulates the values of Table (2) in Equation (IV) to get,
Thus, the specific entropy at state 9 is,
Refer Table A-13, “superheated refrigerant 134a”, and write the specific enthalpy at state 4 corresponding to pressure at state 4 of
Substitute
Hence, the amount of heat removed from the refrigerated space is
Substitute
Hence, the compressor work per unit mass of refrigerant flowing through the condenser is
(c)
The coefficient of performance of the system.
Answer to Problem 113RP
The coefficient of performance of the system is
Explanation of Solution
Express the coefficient of performance of the system.
Conclusion:
Substitute
Hence, the coefficient of performance of the system is
Want to see more full solutions like this?
Chapter 11 Solutions
EBK THERMODYNAMICS: AN ENGINEERING APPR
- Why is truck refrigeration designed to provide low- and medium-temperature refrigeration?arrow_forwardHow will oversizing the buffer tank on a water-to-water heat pump system affect the run time of the system?arrow_forwardConsider a two-stage compression refrigeration system operating between the pressure limits of 0.8 and 0.14 MPa. The working fluid is refrigerant-134a. The refrigerant leaves the condenser as a saturated liquid and is throttled a flash chamber operating at 0.32 MPa. Part of the refrigerant evaporates during this flashing process, and this vapor is mixed with the refrigerant leaving the low-pressure compressor. The mixture is then compressed to the condenser pressure by the high-pressure compressor. The liquid in the flash chamber is throttled to the evaporator pressure and cools the refrigerated space as it vaporizes in the evaporator. Assuming the refrigerant leaves the evaporator as a saturated vapor and both compressors are isentropic, determine (a) the fraction of the refrigerant that evaporates as it is throttled to the flash chamber, (b) the amount of heat removed from the refrigerated space and the compressor work per unit mass of refrigerant flowing through the condenser.arrow_forward
- A heat pump operates on the ideal vapor-compression refrigeration cycle and uses refrigerant-134a as the working fluid. The condenser operates at 1400 kPa and the evaporator at 140 kPa. Determine this system's COP and the rate of heat supplied to the evaporator when the compressor consumes 7 kW. (Take the required values from saturated refrigerant-134a tables.) The COP of the system is and the rate of heat supplied to the evaporator is KM 4arrow_forwardDetermine the degrees of subcooling at the exit of the condenser of a 2-ton air-conditioner system. The system operates on the ideal, vapor-compression refrigerationcycle with the following design parameters: R-134a flow rate 0.05 kg/s Evaporator Pressure 200 kPa Condenser Pressure 1200 kPaarrow_forwardA Carnot refrigeration cycle uses refrigerant-134a as the working fluid. The refrigerant evaporates at 120 kPa and condenses at 30ᵒC. The refrigerant changes from saturated vapor to saturated liquid in the condenser as it rejects heat. Determine the heat removal (kJ/kg) from the refrigerated space.arrow_forward
- Consider a two-stage cascade refrigeration system operating between the pressure limits of 1.2 MPa and 200 kPa with refrigerant-134a as the working fluid. The refrigerant leaves the condenser as a saturated liquid and is throttled to a flash chamber operating at 0.32 MPa. Part of the refrigerant evaporates during this flashing process, and this vapor is mixed with the refrigerant leaving the low-pressure compressor. The mixture is then compressed to the condenser pressure by the high-pressure compressor. The liquid in the flash chamber is throttled to the evaporator pressure and cools the refrigerated space as it vaporizes in the evaporator. The mass flow rate of the refrigerant through the low-pressure compressor is 0.11 kg/s. Assuming the refrigerant leaves the evaporator as a saturated vapor and the isentropic efficiency is 85 percent for both compressors, determine (a) the mass flow rate of the refrigerant through the high-pressure compressor, (b) the rate of heat removal from the…arrow_forwardai 10roh Consider a two-stage cascade refrigeration system operating between the pressure limits of 1.2 MPa and 200 kPa with refrigerant-134a as the working fluid. The refrigerant leaves the condenser as a saturated liquid and is throttled to a flash chamber operating at 0.32 MPa. Part of the refrigerant evaporates during this flashing process, and this vapor is mixed with the refrigerant leaving the low-pressure compressor. The mixture is then compressed to the condenser pressure by the high-pressure compressor. The liquid in the flash chamber is throttled to the evaporator pressure and cools the refrigerated space as it vaporizes in the evaporator. The mass flow rate of the refrigerant through the low-pressure compressor is 0.11 kg/s. Assuming the refrigerant leaves the evaporator as a saturated vapor and the isentropic efficiency is 85 percent for both compressors, determine (a) the mass flow rate of the refrigerant through the high-pressure compressor, (b) the rate of heat removal…arrow_forward2. A cascade refrigeration system using R - 22 in the low - temperature circuit and ammonia (R-717) in the high - temperature circuit has a load of 150 kW. The low - temperature circuit operates at an evaporating temperature of -50°C and a condensing pressure of 500 kPa. Refrigerant leaves the low temperature evaporator as saturated vapor and enters the suction of the low - temperature compressor at - 45°C. Liquid refrigerant exits the condenser at 02°C. The high temperature circuit - operates at an evaporating temperature of -10°C and a condensing pressure of 1200 kPa. The refrigerant exits both evaporator and condenser at saturated conditions. Calculate the power input to the high temperature circuit - compressor, kW.arrow_forward
 Refrigeration and Air Conditioning Technology (Mi...Mechanical EngineeringISBN:9781305578296Author:John Tomczyk, Eugene Silberstein, Bill Whitman, Bill JohnsonPublisher:Cengage Learning
Refrigeration and Air Conditioning Technology (Mi...Mechanical EngineeringISBN:9781305578296Author:John Tomczyk, Eugene Silberstein, Bill Whitman, Bill JohnsonPublisher:Cengage Learning
