
Interpretation:
To draw two different three-dimensional representations for CH2Cl2 (dichloromethane) using wedges and dashes
Concept introduction:
In the stereochemistry, various methods are depicted to represent three dimensional representations. Methane, a tetrahedral model, substituted by four different or same types of ligands at the vertices of the tetrahedron, which is not apparent in the
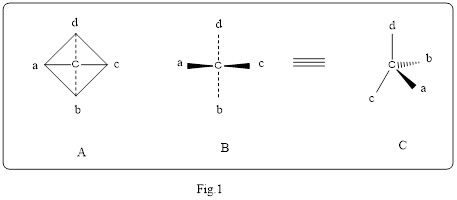
Wedge or solid heavy line represent a bond that project above the plane or pointing towards the observer. Broken wedge or dashed lines are that project below the plane or pointing away from the observer. The wedge and dashed lines for the sample methane is shown below in the Fig.1. Structure A and B represent a normal structure. Structure C represent the wedge and dashed lines. In the Structure C, a represent wedge, b represent dashed lines and c, d represent solid lines.
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
Organic Chemistry
- Select the bonds in the 3D representation of methyl urethane below that are multiple bonds. (Hint: Consider the number of bonds attached to an atom in the image compared to the normal valence of that atom. In a valid structure, the number of bonds to an atom will equal the valence of that atom.) • Gray = C: white = H; red = 0; blue = N; dark green = Cl; brown = Br; light green = F; purple = 1; yellow = S: orange = P. • Double click to select bonds. You can zoom in and out using the mouse scroll wheel (or pinch to zoom on touch screens). ●arrow_forwardSets 1 and 3 are not important, I already figured that part out. But I want to figure out how to explain why LiCl is soluble(water not specified) without using the solubility rule. I believed it to be something to do with higher polarization but I'm not sure.arrow_forwardThere are two substances with the molecular formula C2H7N. Draw them and describe how they differ.arrow_forward
- Butane is rotated along its central carbon bond (C2-C3). What would butane look like if it is rotated 30 degrees? What would butane look like if it is rotated 90 degrees? What would butane look like if it is rotated 150 degrees? Please draw these forms of butane using the wedge and dash method.arrow_forwardwhy does n = t1/t2?arrow_forwardb) H3C ОН NO₂ Sn2 H (+)arrow_forward
- Using the knowledge established in the previous parts, answer the original question: Ultraviolet light with a wavelength longer than about 295 nm has little germicidal value. What is the frequency (in Hz) that corresponds to this wavelength? Hzarrow_forwardC2H2 is acetylene. Apply this changes on the problem when solving. Solve completely.arrow_forwardChemistry 500 A Q2) 1 2 1 pick one. 2 3 4 All or none per row 1 to 7: (spelling counts) Monosodium glutamate (MSG) Center #electron regions (steric units) 5 6 3 S Legend: sodiumoxygen atom(red); carbon atom(black); nitrogen atom(blue); hydrogen atom(white). 7 Assignment #6 VSEPR large molecules: SEPR worksh... 6 #lone electron pairs ★, #bonded electron regions 4 electron geometry EG TE X # T 2023 500 Q6 V... molecular geometry MGH THE THUN Watch BUTTE var 100 M H 156 16 04105 wear ch Herale della COMEarrow_forward
- Predict the MAJOR product of the following reaction sequence. (A EtO OEt HO 1) NaOEt in EtOH 2) BrCH₂CH₂CH₂CH3 3) NaOEt in EtOH 4) allyl bromide 5) H₂O*, heat CH₂ CH3 :arrow_forwardConsider the molecule of chloroform. Its tetrahedral structure is shown. H | CI CCI CI Is this molecule polar? If yes, consider which side of the molecule has partial negative charge and which - partial positive. Then, indicate the direction of the dipole moment relative to the molecule as shown. Non-polar, zero dipole moment. Polar. The dipole vector points along one of the C-Cl bonds. Polar. The dipole vector points along the vertical axis. parallel to the C-H bond. Polar. The dipole vector points along the horizontal axis. Polar. The dipole vector is perpendicular to the plane of the figure.arrow_forwardWhich is the correct 3D representation of lactic acid or are they just the same?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

