
General, Organic, and Biological Chemistry
7th Edition
ISBN: 9781285853918
Author: H. Stephen Stoker
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
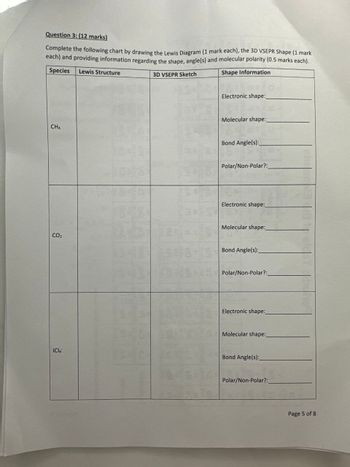
Transcribed Image Text:Question 3: (12 marks)
Complete the following chart by drawing the Lewis Diagram (1 mark each), the 3D VSEPR Shape (1 mark
each) and providing information regarding the shape, angle(s) and molecular polarity (0.5 marks each).
Species Lewis Structure
3D VSEPR Sketch
Shape Information
CH4
Electronic shape:
Molecular shape:
Bond Angle(s):
Polar/Non-Polar?:
Electronic shape:_
CO₂
Molecular shape:
Bond Angle(s):
Polar/Non-Polar?:
Electronic shape:
Molecular shape:_
ICIA
Bond Angle(s):
Polar/Non-Polar?:_
Page 5 of 8
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 2 steps with 3 images

Knowledge Booster
Similar questions
- For each of the following molecular models, write an appropriate Lewis formula.arrow_forwardGiven the bonds C N, C H, C Br, and S O, (a) which atom in each is the more electronegative? (b) which of these bonds is the most polar?arrow_forward3-106 Consider the structure of Penicillin G shown below, an antibiotic used to treat bacterial infections caused by gram-positive organisms, derived from Penicillium fungi: (a) Identify the various types of geometries present in each central atom using VSEPR theory. (b) Determine the various relative bond angles associated with each central atom using VSEPR theory (c) Which is the most poiar bond in Penicillin G? (d) Would you predict Penicillin G to be polar or nonpolar?arrow_forward
- Use the Molecule Shape simulator (http://openstaxcollege.org/I/6MolecShape) to build a molecule. Starting with the central atom, click on the double bond to add one double bond. Then add one single bond and one lone pair. Rotate the molecule to observe the complete geometry. Name the electron group geometry and molecular structure and predict the bond angle. Then click the check boxes at the bottom and right of the simulator to check your answers.arrow_forward7.30 The bond in HF is said to be polar, with the hydrogen carrying a partial positive charge. For this to be true, the hydrogen atom must have less than one electron around it. Yet the Lewis dot structure of HF attributes two electrons to hydrogen. Draw a picture of the electron density distribution for HF and use it to describe how the hydrogen atom can carry a partial positive charge. How can these two models of the HF bond (the electron density and the Lewis structure) seem so different and yet describe the same thing?arrow_forwardhat does it mean to say that a bond is polar? Give two examples of molecules with polar bonds. Indicate in your examples the direction of the polarity.arrow_forward
- Fill in the blanks in each line of the following table that involves characteristics of various bonds between nonmetals. The first line is already completed as an example.arrow_forwardThe connectivity of HNO could be either HNO or HON. Draw a Lewis structure for each and predict which connectivity is the more favorable arrangement.arrow_forwardSuccessive substitution of F atoms for H atoms in the molecule NH3 produces the molecules NH2F, NHF2, and NF3. a. Draw Lewis structures for each of the four molecules. b. Using VSEPR theory, predict the geometry of each of the four molecules. c. Specify the polarity (polar or nonpolar) for each of the four molecules.arrow_forward
- The percent ionic character of a bond can be approximated by the formula 16+3.52 , where is the magnitude of the difference in the electronegativities of the atoms (see Fig. 3.18). Calculate the percent ionic character of HF, HCl, HBr, HI, and CsF, and compare the results with those in Table 3.7.arrow_forward3-105 Consider the structure of Vitamin E shown below, which is found most abundantly in wheat germ oil, sunflower, and safflower oils: (a) Identify the various types of geometries present in each central atom using VSEPR theory. (b) Determine the various relative bond angles as sociated with each central atom using VSEPR theory. (c) Which is the most polar bond in Vitamin E? (d) Would you predict Vitamin E to be polar or nonpolar?arrow_forwardSuccessive substitution of F atoms for H atoms in the molecule CH4 produces the molecules CH3F, CH2F2, CHF3, and CF4. a. Draw Lewis structures for each of the five molecules. b. Using VSEPR theory, predict the geometry of each of the five molecules. c. Specify the polarity (polar or nonpolar) for each of the five molecules.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning


Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning