Q: How many moles of Na,SO4 are in 100.0 mL of a 0.2500 M solution ? The GFW is 142.07 g. O A. 0.0500…
A: 0.25 M Na2SO4 solution. Molarity = number of moles in 1L of solution So, 0.25 moles of Na2SO4 in 1 L…
Q: 0.0050 M KOH PH VALUE
A: KOH stands for potassium hydroxide. It is an inorganic compound with the chemical formula of KOH.
Q: How many ml 2M NaOH would it take to neutralize 20 ml of 0.5M HCI?
A: Given: Concentration of NaOH , M1 = 2 M Volume of NaOH, V1 =? Concentration of HCl, M2 = 0.5 M…
Q: BSA mg/ml O.D. 550nm 1 0.12 2 0.23 3 0.34 4 0.41 5 0.52 Use the values above to draw a graph
A: BSA Bovine serum albumin is a albumin protein which is extracted from cow and used in experimental…
Q: How many grams is one mole of potassium chloride KCI?
A: Moles : unit of concentration Mole concept is used to convert mass and number of particles. 1 mole…
Q: How many milliliters of Children’s Motrin (100 mg of ibuprofen per 5 mL) are needed to give a child…
A: Ibuprofen is a medication in the non-steroidal anti-inflammatory drug (NSAID) that is used to treat…
Q: Define buffer.
A: An aqueous solution that comprises of a weak acid and its conjugate base's mixture or a weak base…
Q: Calculate the amount of dextrose and sodium chloride in 750 mL of D5NS.
A: DSNS solution is Dextrose Sodium Chloride Solution. 5% Dextrose and 4.5% Sodium Chloride is normally…
Q: The doctor ordered Claforan 1400mg IM every 12 hours. The directions for the 2 gm vial state: Add…
A: The doctor ordered Claforan 1400mg IM every 12 hours. The directions for the 2 gm vial state: Add…
Q: The vapour pressure of water at blood temperature is 47 Torr. What is the partial pressure of dry…
A: Vapour pressure is a measure of the tendency of a material to change into the gaseous or vapour…
Q: 50 mL of 5mM NaCl solution from 1 M NaCl solution
A: Dilution is the process by which we decrease the concentration of a solution. This process is vital…
Q: What is the relationship betweenpKa and the useful range of a buffer?
A: The strength of an acid in a solution can be expressed in terms of the acid dissociation constant…
Q: When measuring the volume of a liquid in a graduated cylinder, at which point should the measurement…
A: A graduated cylinder is used to meausre the volume of liquids. Liquids that are to be measured can…
Q: Which of the following is not an equivalent to ppm? a.mg/L b.ug/g c.g/ton d.uL/mL x 106
A: The abbreviation of ppm is ‘parts per million'. It is also expressed as milligrams per litre (mg/L).…
Q: our patient drank the following liquids for breakfast;220 ml of orange juice,180 ml of milk and 150…
A: Nutrition is a process in which an organism uses food to support its life as food provides energy…
Q: 8.5gm/250ml = (x)gm/630ml
A: The amount of chemical dissolved in the solvent = 8.5gm The volume of the solvent…
Q: Determine the mass of baking soda (84 g/mol) needed to completely react with 531mL of vinegar.…
A: Acid and base react to produce salt and water. In some reactions, gas may also be released (hydrogen…
Q: How many moles of tungsten(VI) oxide and hydrogen were required to produce the 5.00 g of tungsten?
A: Stoichiometry is a section of chemistry that involves using relationships between reactants and/or…
Q: If 4.00 L of ethyl alcohol at 18.0 ⁰C is exposed to a temperature of 25.0 ⁰C, what is the resulting…
A: Initial temparature of ethyl alcohol is T₁ = 18° C = (273 +18) K = 291 K rising the temparatute to…
Q: 00 ml total solution, the resulting stren 5..?
A: A pharmaceutical medicine is a chemical substance that causes a biological or physiological effect…
Q: If a 1.20 L of strawberry wine is 10.5% alcohol by volume, how many ml of the solution is ethanol?
A: Wine is an alcoholic beverage produced from honey, fruits like grapes, pineapple, strawberry, etc.…
Q: How many grams are in 0.250 moles of lithium carbonate ? a) 12.225 b) 18.47 c) 18.5 d) 16.5
A: The molecular formulae of lithium carbonate are Li2CO3.
Q: What is 10.2ml in liquid volume?
A: When measuring the volume of a liquid, sometimes referred to as capacity, the units litres (L) and…
Q: How many milliliters of 0.1M aqueous Sodium Hydroxide solution can be prepared from 10 gm ?of Sodium…
A: The molarity (M) is the amount of a substance dissolved in a certain volume of a solution. It is…
Q: What is the volume (in mL) of ethanol in the bottle?
A: Volume is calculated by the % by volume formula, that is volume of solute dissolved in the volume of…
Q: Calculate the volume of TAE buffer that you will need to prepare a 100ml solution of 1X strength…
A: TAE buffer stands for Tris-acetate-EDTA buffer. It is used in lab to do electrophoresis of nucleic…
Q: h thiopental ar d in the lethal
A: Thiopental and pentobarbital used in anesthesia, after rapid intravenous injection of a single dose…
Q: determine the density of a metal that has a mass of 2.17g and a volume of 0.35 cm^3
A: The unit of density is gm/cm3 .
Q: A solution is made by mixing 56.9 g of water and 23.8 g NaCl. What is the concentration of NaCl in…
A: Given Values: Weight of the water = 56.9 grams Weight of NaCl = 23.8 grams
Q: 2.2 Assuming that the final volume of the solution after the titration is exactly 5 mL, determine…
A: Molarity(M) = [moles of solute(n)]÷[litres of solution (v)] Atomic mass of sulpher= 32+(16×4) =96.…
Q: Find the number of moles for acetylsalicylic acid when the mass is 5.08g.
A: Also known as Aspirin, acetylsalicylic acid (ASA) is a usually a drug used for the treatment of pain…
Q: the vapor phase concentration in ppm(v)?
A: Vapour phase concentration is the amount of water vapor present in a unit volume of air, usually…
Q: Mixing 50.0 mL of 0.200 M Na2HPO4 with 50.0 mL of 0.120 M HCI results in a buffer solution composed…
A: HCl is a strong acid. It is highly corrosive
Q: Why should Erlenmeyer flasks hold not more than 60% of their capacity?
A: To conduct experiments and conduct research, laboratories need a variety of instruments and…
Q: Calculate the pH of the solution that results following addition of 10 mL of 1 M NaOH to 40 mL of 1…
A: Henderson-Hasselbalch equation gives the pH of the solution and relates to the concentration of…
Q: 2 mcg/kg/min Pt weighs 165 lbs. Drug 50mg in 1000mL. How many mL/hr.?
A: When order is written in units mcg/kg/min the following equation is used wieght can be converted to…
Q: Object A has a mass of 40g and a volume of 30mL and object B has a mass of 15 g and a volume of 20…
A: Ans 30. Density is the mass per volume measurement. The average object density refers to its total…
Q: Determine the number of moles in 1.95 x 1023 formula units of MgCl2.
A: Given: Formula units of MgCl2=1.95×10233 No. of moles of MgCl2=?
Q: How many moles of HCl are there in 75.0 mL of 0.160 M HCl?
A: Molarity can be stated as the soluble moles of solute, which is dissolved per liter of solution.
Q: 9% dextrous solution (9 mg per 100 ml. of solution) is given
A: A. 1a. In order to calculate flow rate of dextrose we have to use following formula 2.18 L = 2180…
Q: alanine that would exist at the pH indicated below.
A:
Q: Calculate the mass of a liquid with a density of 3.2 g/mL and a volume of 25 mL
A: The amount of matter present in a substance is defined as its mass. It is independent of its…
Q: How many liters of a 7 M NaOH stock solution would you need to make 780 mL of a 153 mM NaOH…
A: Molarity is defined as the number of moles of solute present in a liter of solution. Molarity =…
Q: If a sukfuric acid solution is titrated with the above sodium hydroxide soultion, it reqires 45.20…
A: Acid-base titration: This is a standard procedure to measure the concentration of unknown acid/base…
Q: ounce 8 = how many drams ?
A: The different systems of measurement used in medicine include the household system, the avoirdupois…
Q: How many mL of a ½ strength magnesium sulfate solution will contain 10 grams of magnesium sulfate?
A: Drug dose is the amount of drug in mg which is needed to be administered into the patient Strength…
Q: GC/L what does the E and GC/L stand for in water measurement?
A: EOP - electro-osmotic-pulse GCL - geosynthetic liners
Q: Using the table above, make the following unit conversions. Determine th point the appropriate…
A: 1. 25/1000 = 0.025 2. 3*1000000= 3000000 3. 150/10 = 15 4. 5000/1000 = 5 5. 0.075 kl
Q: What does “1 Millimolar” (1mM) mean
A: Milli is a suffix in measurement meaning 1/1000 or 10^-3.
Convert 12 pints to an equivalent volume in quarts.
- In USA there's a dry pint and a liquid pint which measure different.Dry pint measures 550.6 ml and a liquid pint measures 473.2 ml.For both dry and liquid pint two cups make one and two pints are equal to one quart.
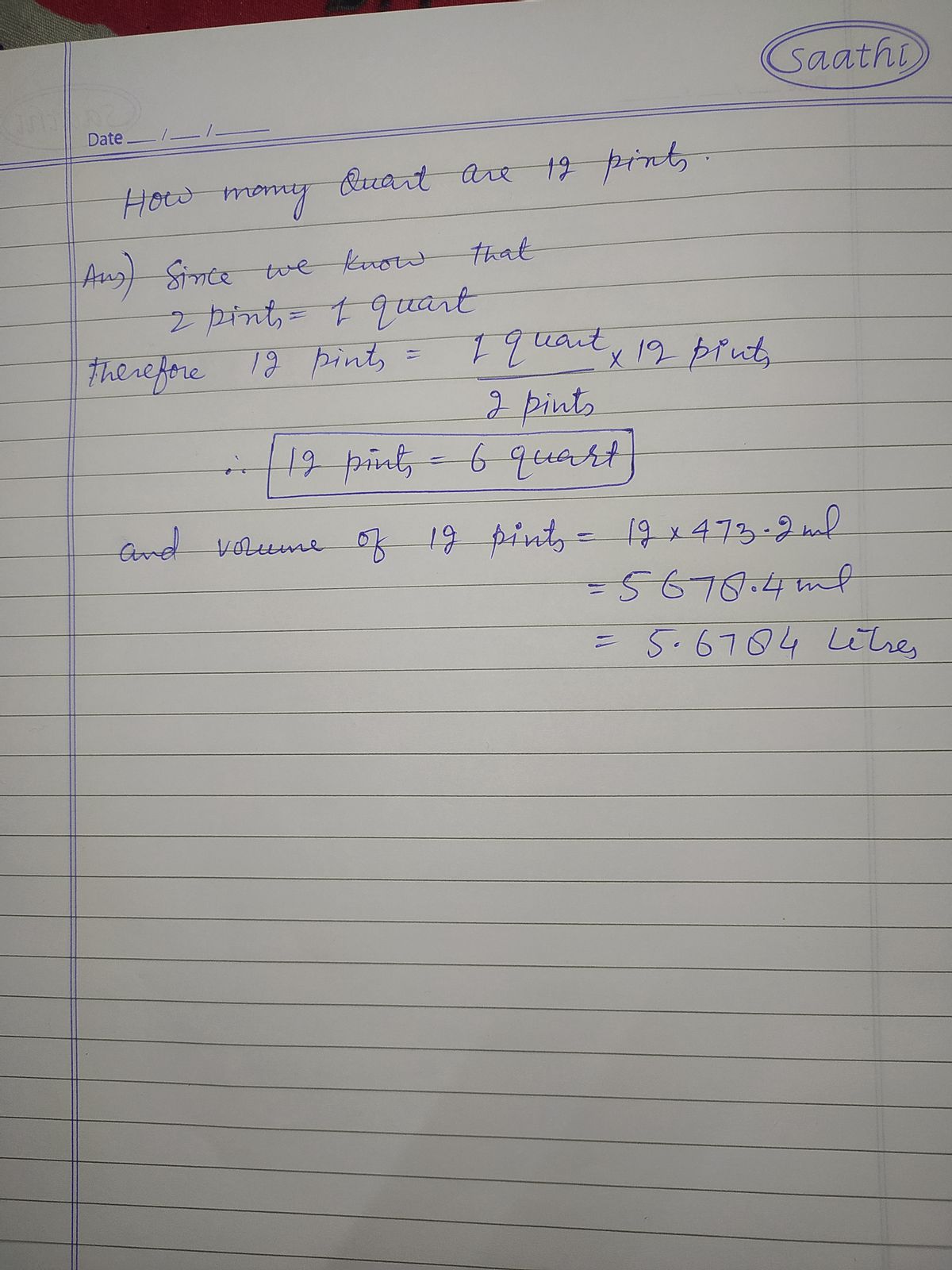
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images










