
Human Anatomy & Physiology (11th Edition)
11th Edition
ISBN: 9780134580999
Author: Elaine N. Marieb, Katja N. Hoehn
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Question

Transcribed Image Text:a) What technique(s) can be used to determine the structure of the oxygen-
evolving complex as shown in A?
b) In the oxygen-evolving complex A, what type of bond connects Ca1
(Ca2+) and 01?
c) In photosynthesis, the oxygen-evolving complex A works as a water-
oxidizing agent which can produce O2 from H2O under sunlight. In this
process, does H20 act as a Lewis acid or Lewis base?
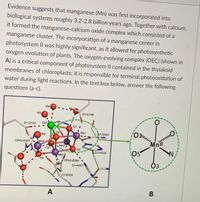
Transcribed Image Text:Evidence suggests that manganese (Mn) was first incorporated into
biological systems roughly 3.2-2.8 billion years ago. Together with calcium,
it formed the manganese-calcium oxide complex which consisted of a
manganese cluster. The incorporation of a manganese center in
photosystem II was highly significant, as it allowed for photosynthetic
oxygen evolution of plants. The oxygen-evolving complex (OEC) (shown in
A) is a critical component of photosystem II contained in the thylakoid
membranes of chloroplasts; it is responsible for terminal photooxidation of
water during light reactions. In the text box below, answer the following
questions (a-c).
W4
W3
D1-E189
D1-D170
Ca1
01 1
05
Mn1
D1-D342
W2
CP43-R357
Mn4
Mn2
03
05
D1-H332
Mn3
CP43-E354
Оз
D1-H337
D1-E333
A
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- We learned that three different amino acid transformations of PLP-dependent enzymes canresult from different conformations of the PLP-amino acid imine adduct in the active site.Starting from the PLP adduct of (S)-serine, show mechanisms fora) Decarboxylation of serineb) Racemization of serinec) Conversion of serine to glycine and formaldehyde.arrow_forward(a) You have found a new, enhanced version of the drug jafrasitor, (called jafrasitorplus) (molecular weight 600 grams/mol), that binds the histone deacetylase enzyme Sir2 with a dissociation constant KD = 0.03 nM. What mass of jafrasitorplus should be administered to a patient with a blood volume of 5.5 L such that Sir2 is at least 91% inhibited? Show your work and reasoning.arrow_forwardExplain how lysyl-tRNA synthetase protein in bacterial cells typically undergoes folding with necessary structures and steps, any ATP hydrolysis, cap structure, etc.arrow_forward
- You generate mutants in the metabolic pathway for starlase. You conduct some complementation tests (after testing for dominance of course) and come up with the following results: 1 2 4 6 1 + + + 2 + 3 + 4 + 5 6 a. How many complementation groups are there? [Select] b. You conduct some additional experiments to elucidate the starlase metabolic pathway. Your results are shown below. Use this information alongside information from the complementation table above to place the intermediates in the correct order on the pathway. (HINT: use the complementation groups from the table above to help you consolidate information on the tables below. Reference practice question 3 from today's lecture for help). Addition to minimal medium Mutant None starlase P 1 + + 2 + + 4 + + 5 + 6 Precursor --> [ Select ] [ Select ] [ Select ] --> starlase c. Mutant 4 has a loss-of-function mutation for which enzyme in the starlase synthesis pathway? [ Select ] E1 E2 ЕЗ Е4 Precursor > Intermediate 1→ Intermediate…arrow_forwarda. A single mutation in the riboswitch’s sequence changes the ligand binding specificity from guanine toadenine. Predict what single mutation could be made in the guanine-responsive riboswitch sequenceto convert it to an adenine-responsive riboswitch. Justify your answer b. Could a DNA molecule with the same nucleotide sequence as the guanine-responsive riboswitch bindguanine in a similar manner as the RNA-based molecule? Explainarrow_forwardin all cells observed to date, DNA serves as the stable, information-rivh molecule that stores genetic information. In contrast, in almost all cases, non-catalytic RNS serves as either a labile intermediate molecule involve in the production of proteins or as a labile regulatory molecule. A) based on the material, hypothesize why the molecule structures of DNA and RNA are, or are not, consistent with these two roles. B) RNA can also serve a catalytic role(ribosomes, RNA splicing, etc). What part of the RNA molecule would you predict makes it more likely than DNA to participate in enzymatic reactions? C) Explain how all these ideas may support as "RNA World Hypothesis" where RNA evolved first, followed by DNA and proteinarrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education