
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
thumb_up100%
can someone determine the equivalence points of these graphs and explain how you got those values? thank you
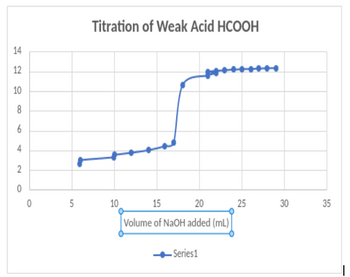
Transcribed Image Text:14
12
10
8
6
4
2
0
0
5
Titration of Weak Acid HCOOH
10
15
20
Volume of NaOH added (mL)
-Series1
25
30
35
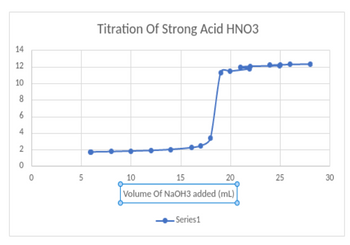
Transcribed Image Text:14
12
10
8
6
4
2
0
0
5
Titration Of Strong Acid HNO3
10
15
20
Volume Of NaOH3 added (ml)
-Series1
25
30
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Think about the situation where you are investigating the relationship of study time vs. exam grade.a. Which variable would be on the x-axis (and why)?b. Which variable would be on the y-axis (and why)?c. Would you expect the slope to be positive or negative (and Why)? 2) Contrast and compare interpolation with extrapolation. Which is “safer” to use and why?3) Look up how “the best fit straight line” is constructed. The term “Least squares” should be in your explanation.4) If I plot some data and end up with a r2 of (-) 0.99, is my plot useless? Explain.arrow_forwardConcentration (mg/L) Ca Mg K Na Al 50.712 68.592 9.357 11.069 12.893 Calculate the CEC for individual soil components and the total CEC for the soil. Place your values in Table 1 below. Table 1. Ca2+ Mg2+ K+ Na+ Al3+ Total Concentration (mg/L) -------- CEC cmol(+)/kgarrow_forward3. Standard white vinegar you can buy in the grocery store is 5% concentration. That means 5% of the liquid vinegar is acetic acid and 95% of the solution is water. In a hardware store, you can buy industrial strength vinegar, which is 30% concentration. This means that 30% of the vinegar is acetic acid, and the remaining 70% is water. Samuel does another experiment, this time with 5% vinegar and 30% vinegar. He sets up two science fair volcanoes (in no particular order), each with the same temperature, mass of baking soda and volume of vinegar. But one volcano uses 5% vinegar and the other volcano uses 30% vinegar. He measures the volume of gas production for the'first minute of each reaction, and he records the data below. Volcano # 1 Time Volume of gas Volume of gas produced (mL) vs. Time (s) for Volcano #1 (s) produced (mL) 60 50 10 20 30 25 38 40 46 30 • Volume of gas produced (ml) 40 50 20 50 52 e 10 60 53 20 40 60 80 Time (s) Volcano # 2 Volume of gas produced (mL) vs. Time (s)…arrow_forward
- 1. (answer the questions in the tan boxes in the tables below each question) A student conducted an investigation to determine the effect of water temperature on the amount of sugar that dissolves in a beaker of water. Identify components for trial 1 of this investigation. Beaker Number 1 2 3 4 Amount of Water (mL) 100 100 100 100 Amount of Water (mL) Trial 1 Temperature of Temperature of Amount of Sugar Sugar (°C) Water (°C) Dissolved (g) 20 20 20 20 Temperature of Sugar 5 10 15 20 185 189 194 204 Temperature of Water Terms Variable Constant Amount of Sugar Dissolvedarrow_forwardMatti is given two bottles of copper (II) sulfate solutions in her senior high chemistry lab. She is told that one bottle contains a saturated solution and the other one contains an unsaturated solution. The solutions are the exact same color and have no particles in the bottles. Describe what Matti can do to distinguish between the two solutions. A- 米 U X2 x? #3 X Next page age You are logged in as Kaylee Krys (Log out) VLL Chemistry CP 2021-2022 (Fickett). Get the mobile app MacBook Air F8 O00 F4 F5 F6 F2 F3 & * @ # $ 7 9 2 3 4 Q W E R T Y А S D F G H M Y Barrow_forwardNeed help on all of them. It is mu first time taking chemistry so I am stuck on what to do. Thank youarrow_forward
- Aluminum-lithium (AI-Li) alloys have been developed by the aircraft industry to reduce the weight and improve the performance of its aircraft. A commercial aircraft skin material having a density of 2.65 g/cm? is desired. Compute the concentration of Li (in wt%) that is required. The densities of aluminum and lithium are 2.71 and 0.534 g/cm3, respectively.arrow_forwardStep 2: Identify the dimensions of the quantities involved The second step is to identify the dimensions of the quantities involved in the problem. For example, if the problem involves distance, time, and velocity, the dimensions of these quantities would be length, time, and length/time, respectively. Step 3: Check if the units cancel out The third step is to check if the units cancel out. To do this, multiply the quantities together and check if the units cancel out, leaving only the desired unit. For example, if you are trying to find the velocity of an object and you know its distance and time, you can multiply distance by time to get velocity. If the units cancel out, you have a physically meaningful result. Step 4: Check if the result makes sense, to do this compare the units of the result with what you would expect based on the Robles statement. Using the step hints above, answer the question. You do not have to solve. Just imagine that you're teaching a friend how…arrow_forwardAn impure sample contains 1.01 g of impurities and 4.92 g of benzoic acid. The impure sample is dissolved in water, then heated. The solution is then cooled. How many grams of benzoic acid will recrystallize when the solution has cooled? Include the unit and two decimal places in your answer. The solubilities of the impurity and benzoic acid in hot water are found as an item in the Recrystallization Experiment folder. Solubility Data for Report Questions 10 Compound Cold Water solubility Hot Water Solubility Benzoic acid 0.42 g per 100 mL of water 5.79 g per 100 mL of water Impurity 0.61 g per 100 mL of water 4.62 g per 100 mL of waterarrow_forward
- Which answer choice best describes a heterogeneous mixture? A solution with different sized particles A solution that exhibits the Tyndall effect Mixtures which have a non-uniform composition Mixtures which have a uniform compositionarrow_forwardAssume the data below was collected for two different sample solutions using the same experimental procedure you carried out. Based on the two lines shown below, which solution (orange data points or blue data points) corresponds to the sample that would look DARKER in terms of how much light goes through the glass. 0.9 y= 4.7713x - 0.0728 R0.99953 0.8 0.7 0.6 0.5 0.4 0.3 0.2 y1.414x - 0.0546 R0.99642 0.1 0.02 0.04 0.06 0.08 0.1 0.12 0.14 0.16 0.18 0.2 0.1 [Concentration, M] orange data points (lower line) blue data points (upper line) Absorbancearrow_forwardWrite balanced equations for the reaction of Al(OH)g with stomach acid (HCI). Express your answer as a chemical equation. Identify all of the phases in your answer. | ΑΣφ ? Submit Request Answer Part B Write balanced equations for the reaction of Mg(OH), with stomach acid (HCI). Express your answer as a chemical equation. Identify all of the phases in your answer.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY