
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Please correct answer and don't use hand rating
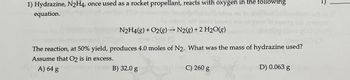
Transcribed Image Text:1) Hydrazine, N2H4, once used as a rocket propellant, reacts with oxygen in the following
equation.
->
N2H4(g) + O2(g) → N2(g) + 2 H2O(g)
The reaction, at 50% yield, produces 4.0 moles of N2. What was the mass of hydrazine used?
Assume that O2 is in excess.
A) 64 g
cal for B) 32.0 g
C) 260 g
D) 0.063 g
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 2 steps

Knowledge Booster
Similar questions
- Chlorine exists mainly as two isotopes, 37Cl and 33Cl. Which is more abundant? How do you know?arrow_forwardA sample of a hydrocarbon (a compound consisting of only carbon and hydrogen) contains 2.59 1023 atoms of hydrogen and is 17.3% hydrogen by mass. If the molar mass of the hydrocarbon is between 55 and 65 g/mol, what amount (moles) of compound is present, and what is the mass of the sample?arrow_forwardAtomic masses are relative masses. What does this mean?arrow_forward
- Nitrogen fixation in the root nodules of peas and other legumes occurs with a reaction involving a molybdenum-containing enzyme named nitrogenase. This enzyme contains two Mo atoms per molecule and is 0.0872% Mo by mass. Calculate the molar mass of the enzyme.arrow_forwardPotassium superoxide, KO2, is employed in a self-contained breathing apparatus used by emergency personnel as a source of oxygen. The reaction is 4KO2(s)+2H2O(l)4KOH(s)+3O2(g) If a self-contained breathing apparatus is charged with 750 g KO2 and then is used to produce 188 g of oxygen, was all of the KO2 consumed in this reaction? If the KO2 wasnt all consumed, how much is left over and what mass of additional O2 could be produced?arrow_forwardThe structure of one of the bases in DNA, adenine, is shown here. Which represents the greater mass: 40.0 g of adenine or 3.0 1023 molecules of the compound?arrow_forward
- The space shuttle environmental control system handles excess CO2 (which the astronauts breathe out; it is 4.0% by mass of exhaled air) by reacting it with lithium hydroxide, LiOH, pellets to form lithium carbonate, Li2CO3, and water. If there are seven astronauts on board the shuttle, and each exhales 20. L of air pee minute, how long could clean air be generated if there were 25,000 g of LiOH pellets available for each shuttle mission? Assume the density of air is 0.0010 g/mL.arrow_forwardopical hydrocortisone is often used to treat a variety of skin conditions, such as insect bites, eczema, and rashes. Each molecule of hydrocortisone contains 21 atoms of carbon (plus other atoms). The mass percentage of carbon in hydrocortisone is 69.5 8%. What is the molar mass of hydrocortisone?arrow_forwardThe active ingredient in Pepto-Bismo® (an over- the-counter remedy for an upset stomach) is bismuth sub-salicylate, C7H5BiO4. Analysis of a 1.7500-g sample of Pepto-Bismol yields 346 mg of bismuth. What percent by mass is bismuth subsalicylate in the sample? (Assume that there are no other bismuth-containing compounds in Pepto-Bismol.)arrow_forward
- Consider the hypothetical reaction 8A2B3(s)+3X4(g)4A4X3(s)+12B2(g)When 10.0 g of A2B3(MM=225g/mol) react with an excess of X4, 4.00 g of A4X3 are produced. (a) How many moles of A4X3 are produced? (b) What is the molar mass of A4X3?arrow_forwardA piece of nickel foil, 0.550 mm thick and 1.25 cm square, is allowed to react with fluorine, F2, to give a nickel fluoride. (a) How many moles of nickel foil were used? (The density of nickel is 8.902 g/cm3.) (b) If you isolate 1.261 g of the nickel fluoride, what is its formula? (c) What is its complete name?arrow_forwardCopper metal reacts with mine acid. Assume that the reaction is 3Cu(s)+8HNO3(aq)3Cu(NO3)2(aq)+2NO(g)+4H2O(l) If 5.58 g Cu(NO3)2 is eventually obtained, how many grams of nitrogen monoxide, NO, would have formed?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning


Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning