
Concept explainers
Interpretation:
Lewis structure for ethylene oxide, ethylene glycol, and acrylonitrile has to be drawn and also the hybrid orbitals of the central atom have to be given along with the number of pi bonds present.
Concept Introduction:
Lewis structure is used for predicting the shape of molecules. From the steric number obtained in a Lewis structure, the molecular geometry can be predicted. VSEPR model can predict the shape of molecules considering their Lewis structure. Certain rules has to be followed in for the VSEPR model.
- The molecule will have a shape where there is minimal electrostatic repulsion between the valence‑shell electron pairs.
- The forces of repulsion between two lone pairs of electrons will be higher than the repulsion between lone pair and bond pair of electrons. This in turn will be higher than the bond pair‑bond pair of electrons.
The hybridized orbitals and the steric number can be related as shown below;
| Steric number | Hybridized orbital |
| 2 | |
| 3 | |
| 4 | |
| 5 | |
| 6 |
Explanation of Solution
Ethylene oxide:
Formula for ethylene oxide is
The total number of valence electrons is calculated as shown below;
A total of
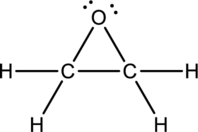
Hybrid orbitals of central carbon atoms:
The carbon atom has does not have a lone pair of electrons and it is bonded to four atoms. Therefore, the steric number is calculated as shown below;
As the steric number is four, the hybridization of carbon atom is
Hybrid orbitals of central oxygen atom:
The oxygen atom has two lone pair of electrons and it is bonded to two atoms. Therefore, the steric number is calculated as shown below;
As the steric number is four, the hybridization of oxygen atom is
There is no unhybridized orbital left out for the formation of pi bonds. Hence, there is no pi bonds in ethylene oxide.
Ethylene glycol:
Formula for ethylene glycol is
The total number of valence electrons is calculated as shown below;
A total of
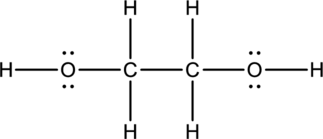
Hybrid orbitals of central carbon atoms:
The carbon atom has does not have a lone pair of electrons and it is bonded to four atoms. Therefore, the steric number is calculated as shown below;
As the steric number is four, the hybridization of carbon atom is
Hybrid orbitals of central oxygen atoms:
The oxygen atom has two lone pair of electrons and it is bonded to two atoms. Therefore, the steric number is calculated as shown below;
As the steric number is four, the hybridization of oxygen atom is
There is no unhybridized orbital left out for the formation of pi bonds. Hence, there is no pi bonds in ethylene glycol.
Acrylonitrile:
Formula for acrylonitrile is
The total number of valence electrons is calculated as shown below;
A total of
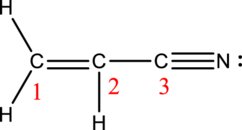
Hybrid orbitals of carbon atom C-1:
The carbon atom has does not have a lone pair of electrons and it is bonded to three atoms. Therefore, the steric number is calculated as shown below;
As the steric number is three, the hybridization of carbon atom is
Hybrid orbitals of carbon atom C-2:
The carbon atom has does not have a lone pair of electrons and it is bonded to three atoms. Therefore, the steric number is calculated as shown below;
As the steric number is three, the hybridization of carbon atom is
Hybrid orbitals of carbon atom C-3:
The carbon atom has does not have a lone pair of electrons and it is bonded to two atoms. Therefore, the steric number is calculated as shown below;
As the steric number is two, the hybridization of carbon atom is
There are one double bond and one triple bond present in the structure. As multiple bonds are present, acrylonitrile contains pi bonds.
Want to see more full solutions like this?
Chapter 10 Solutions
Chemistry: Principles and Practice
- A toxic cloud covered Bhopal, India, in December 1984 when water leaked into a tank of methyl isocyanate, and the product escaped into the atmosphere. Methyl isocyanate is used in the production of many pesticides. Draw the Lewis structures for methyl isocyanate, CH3NCO, including resonance forms. The skeletal structure isarrow_forwardSulfuric acid is the industrial chemical produced in greatest quantity worldwide. About 90 billion pounds are produced each year in the United States alone. Write the Lewis structure for sulfuric acid, H2SO4, which has two oxygen atoms and two OH groups bonded to the sulfur.arrow_forwardBond Enthalpy When atoms of the hypothetical element X are placed together, they rapidly undergo reaction to form the X2 molecule: X(g)+X(g)X2(g) a Would you predict that this reaction is exothermic or endothermic? Explain. b Is the bond enthalpy of X2 a positive or a negative quantity? Why? c Suppose H for the reaction is 500 kJ/mol. Estimate the bond enthalpy of the X2 molecule. d Another hypothetical molecular compound, Y2(g), has a bond enthalpy of 750 kJ/mol, and the molecular compound XY(g) has a bond enthalpy of 1500 kJ/mol. Using bond enthalpy information, calculate H for the following reaction. X2(g)+Y2(g)2XY(g) e Given the following information, as well as the information previously presented, predict whether or not the hypothetical ionic compound AX is likely to form. In this compound, A forms the A+ cation, and X forms the X anion. Be sure to justify your answer. Reaction: A(g)+12X2(g)AX(s)The first ionization energy of A(g) is 400 kJ/mol. The electron affinity of X(g) is 525 kJ/mol. The lattice energy of AX(s) is 100 kJ/mol. f If you predicted that no ionic compound would form from the reaction in Part e, what minimum amount of AX(s) lattice energy might lead to compound formation?arrow_forward
- Nitrosyl azide, N4O, is a pale yellow solid first synthesized in 1993. Write the Lewis structure for nitrosyl azide.arrow_forwardThe study of carbon-containing compounds and their properties is called organic chemistry. Besides carbon atoms, organic compounds also can contain hydrogen, oxygen, and nitrogen atoms (as well as other types of atoms). A common trait of simple organic compounds is to have Lewis structures where all atoms have a formal charge of zero. Consider the following incomplete Lewis structure for an organic compound called histidine (an amino acid), which is one of the building blocks of proteins found in our bodies: Draw a complete Lewis structure for histidine in which all atoms have a formal charge of zero.arrow_forwardDraw a Lewis structure for each of the following molecules or ions. (a) CS2 (b) BF4 (c) HNO2 (where the bonding is in the order HONO) (d) OSCl2 (where S is the central atom)arrow_forward
- Draw Lewis structures for the following species. (The skeleton is indicated by the way the molecule is written.) (a) Cl2CO (b) H3C—CN (c) H2C—CH2arrow_forwardIn which of the following molecules does the sulfur have an expanded octet? For those that do, write the Lewis structure. (a) SO2 (b) SF4 (c) SO2Cl2 (d) SF6arrow_forwardDraw Lewis structures for the following species. (The skeleton is indicated by the way the molecule is written.) (a) Cl2CO (b) H3CCNarrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning





