
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6.SE, Problem 27EDRM
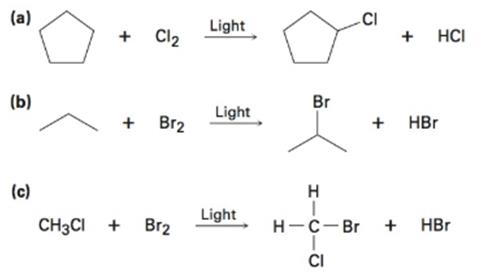
Draw the electron-pushing mechanism for each radical reaction below. Identify each step as initiation, propagation, or termination.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
4) Draw the products following a radical bromination. Identify radicals, and draw
resonance structures.
Ph
NBS/peroxide bromination
Radical reactions
Draw the products formed in each reaction.
The following molecules are subject to substitution (SN1 or SN2) reaction conditions.
a)
Identify if the leaving group (-Br) is attached to a 3°, 2°, 1°, or methyl Carbon.
b)
Rank the molecule with respect to their SN1 reactivity, with 1 being the fastest and 4 being the slowest.
c)
Rank the molecule with respect to their SN2 reactivity, with 1 being the fastest and 4 being the slowest.
Br
Br
CH3B
Br
a. type of Carbon
on C-Br
b. SN1 reactivity
(1 fastest, 4 slowest)
c. SN2 reactivity
(1 fastest, 4 slowest)
Chapter 6 Solutions
Organic Chemistry
Ch. 6.1 - Prob. 1PCh. 6.3 - Prob. 2PCh. 6.3 - Using curved fishhook arrows, propose a mechanism...Ch. 6.4 - Prob. 4PCh. 6.4 - An electrostatic potential map of boron...Ch. 6.5 - What product would you expect from reaction of...Ch. 6.5 - Reaction of HBr with 2-methylpropene yields...Ch. 6.6 - Prob. 8PCh. 6.6 - Predict the products of the following polar...Ch. 6.7 - Which reaction is more energetically favored, one...
Ch. 6.7 - Prob. 11PCh. 6.9 - Which reaction is faster, one with ∆G‡ = +45...Ch. 6.10 - Prob. 13PCh. 6.SE - Prob. 14VCCh. 6.SE - Prob. 15VCCh. 6.SE - Prob. 16VCCh. 6.SE - Look at the following energy diagram: (a) Is...Ch. 6.SE - Look at the following energy diagram for an...Ch. 6.SE - What is the difference between a transition state...Ch. 6.SE - Prob. 20EDRMCh. 6.SE - Prob. 21EDRMCh. 6.SE - Draw an energy diagram for a two-step exergonic...Ch. 6.SE - Draw an energy diagram for a reaction with keq =...Ch. 6.SE - The addition of water to ethylene to yield ethanol...Ch. 6.SE - When isopropylidenecyclohexane is treated with...Ch. 6.SE - Prob. 26EDRMCh. 6.SE - Draw the electron-pushing mechanism for each...Ch. 6.SE - Draw the complete mechanism for each polar...Ch. 6.SE - Prob. 29EDRMCh. 6.SE - Identify the functional groups in the following...Ch. 6.SE - Identify the following reactions as additions,...Ch. 6.SE - Identify the likely electrophilic and nucleophilic...Ch. 6.SE - For each reaction below identify the electrophile...Ch. 6.SE - Prob. 34APCh. 6.SE - Follow the flow of electrons indicated by the...Ch. 6.SE - Prob. 36APCh. 6.SE - Prob. 37APCh. 6.SE - Despite the limitations of radical chlorination of...Ch. 6.SE - Prob. 39APCh. 6.SE - Answer question 6-39 taking all stereoisomers into...Ch. 6.SE - Prob. 41APCh. 6.SE - Prob. 42APCh. 6.SE - Prob. 43APCh. 6.SE - The reaction of hydroxide ion with chloromethane...Ch. 6.SE - Prob. 45APCh. 6.SE - Ammonia reacts with acetyl chloride (CH3COCl) to...Ch. 6.SE - The naturally occurring molecule α-terpineol is...Ch. 6.SE - Prob. 48APCh. 6.SE - Prob. 49APCh. 6.SE - Draw the structures of the two carbocation...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the electron-pushing mechanism for the propagation steps of the allylic bromination reactions below. You may omit NBS in your mechanism, and use Br and Br2.arrow_forwardIdentify the nucleophile and leaving group and draw the products of each substitution reaction.arrow_forwardd. HBr di-tert-butyl peroxide hv or A ndations of Organic Chemistry (Volume 1arrow_forward
- Allylic bromination of alkenes is accomplished with the reagent NBS (N-bromosuccinimide). The reaction proceeds by a radical chain mechanism. For the following reaction: NBS Br light, CCI4 a Select the species that you would expect to react in the first propagation step and draw curly arrows to show the mechanism. Use half-headed (fishhook) curved arrows to show electron reorganization. When drawing bond formation by pairing of single electrons, terminate the arrows in a hotspot located to one side of a reacting radical. This hotspot is not associated with any structural feature. Arrow-pushing Instructions 将一郎: Br H2 Previous Nextarrow_forward6) Write the mechanism (two propagation steps only, starting with the bromine radical in the first step and using Br2 in the second step) and that would explain how the following two products are produced. (Hint – the allylic radical is resonance stabilized.) Br Br NBSarrow_forward4 VI. Draw the each step of the mechanism for free-chain radical reaction of butane (include the primary and secondary positions).arrow_forward
- How might nucleophilic catalysis work?Draw out a possible mechanism.arrow_forward5a) Draw the product(s) of the reaction shown below. Additionally, identify the nucleophile, electrophile and leaving group.arrow_forwardAs we will learn, many antioxidants–compounds that prevent unwanted radical oxidation reactions from occurring–are phenols, compounds that contain an OH group bonded directly to a benzene ring.a.) Explain why homolysis of the O–H bond in phenol requiresconsiderably less energy than homolysis of the O–H bond in ethanol(362 kJ/mol vs. 438 kJ/mol).b.) Why is the C–O bond in phenol shorter than the C–O bond in ethanol?arrow_forward
- Which reagent(s) adds HBr to a double bond in a non-Markovnikov fashion via a radical mechanism? A. Br2 B. Br2, hv or light C. HBr D. HBr with CH3OOCH3arrow_forward4. The following radical reaction produces 3 unique products. Draw the products AND SHOW YOUR WORK (arrows). NBS, hv 3 products H3C `CH3arrow_forwardPlease answer this organic chemistry question: show all three steps and radical mechanism for hydrobromic acid in peroxide reacting with 3-methylene-2-ene. Are there multiple products? Background information just in case needed for the intial question above ? but if not needed just ignore this background information: which of the following compounds will react the quickest? A) Fluorine with a tertiary radical B) chlorine with a tertiary radical C) Bromine with tertiary radical D) chlorine with a primary radical E) bromine with a primary radicalarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY