Q: What cause the spikes in the spectrophotometer reading? Explain
A: In electronic spectroscopy, generally the electron excites from an initial low energy state to a…
Q: If the TLC plates had been developed in a more polar solvent, would retention factors on the whole…
A:
Q: Calculate the Re value for a spot in a TLC experiment if the solvent moved 14.5 cm and the spot…
A: Given Distance moved by solvent = 14.5 cm Spot of Compound moved from the origin = 6.1 cm Rf =…
Q: Calculate the Rf value for a spot in a TLC experiment if the solvent moved 14.9 cm and the spot…
A: Given values, Distance travelled by solvent = 14.9 cm Distance travelled by solute= 9.5 cm
Q: Consider the retention time of a pesticide is.13 8.5 min and the peak width at the base = 1.7 min,…
A: Given that, The retention time of a pesticide tr = 8.5 min Peak width at the base σt = 1.7 min…
Q: A student performs this and records the following data from the complted chromatogram. the solvent…
A: The Rf value is the ratio of distance travelled by the sample (b) to the distance travelled by the…
Q: An analgesic drug sample was analyzed using TLC to determine its probable active ingredient. The…
A: TLC is thin layer chromatography technique which is used to separate the mixture of non-volatile…
Q: Which of the following methods cannot be used to visualize spots on a TLC plate? Group of answer…
A: Which of the following methods cannot be used to visualize spots on a TLC plate? Group of answer…
Q: What is the Rf value for the uppermost yellow spot in TLC plate 3 (plate imaged below)? It is…
A: The TLC plate 3 with all the spots is given below :
Q: A spot of black ink was placed on a strip of filter paper. The chromatogram was developed using an…
A: The fastest moving spot has the highest Rf value. here we see C covered a long distance so it has…
Q: What adsorbents are typically used in TLC? On a TLC, a compound traveled 3.5 cm when the solvent…
A: Thin layer chromatography is the technique that is used to separate or purify the compound from a…
Q: How can you vary the retention factor and selectivity factor?
A: Retention factor is a variable which indicates the time duration a component spends in the…
Q: analgesic drug sample was analyzed using TLC to determine its probable active ingredient. The…
A:
Q: Which technique measures secondary structure? a. Size exclusion chromatography b. Circular Dichroism…
A: The secondary structure is formed by bending of the 1o structure. In this, an intermolecular force…
Q: Calculate the Rf value for a spot in a TLC experiment if the solvent moved 13.8 cm and the spot…
A: In the given TLC experiment the movent of the solvent is = 13.8 cm The movement of the spot is = 6.5…
Q: A student measures the solvent height on a TLC plate as 11.5 cm. Spot A was measured at a height of…
A: Formula used :- RF = distance traveled by compound/distance traveled by solvent
Q: If a spot travels 2.3 cm while the mobile phase travels 6.6 cm, what is the Rf value of the…
A: Given: Distance travelled by spot = 2.3 cm Distance travelled by mobile phase = 6.6 cm
Q: Before developing a TLC plate you should _______. a. Make sure to mark the origin with a pen b.…
A: While developing a TLC plate, we should always use a pencil (not pen) because: Pencils are cheap and…
Q: Why is it that a warm cuvette does not lose any significant heat during the absorbance measurement…
A: Cuvettes are designed to hold samples specially for spectroscopic measurement. Here the light beam…
Q: spot of black ink was placed on a strip of filter paper. The chromatogram was developed using an…
A:
Q: Why should you not use a ballpoint pen when marking a TLC plate and why does the horizontal line…
A: If ball point pen is used, the ink will travel up with the solvent, just like the samples. The…
Q: What is the design of figure II? FT IR interferometer single beam spectrophotometer…
A: The design of figure II is
Q: 13. Consider the TLC plate below to answer the question. What is the Rf of the top component? Note:…
A: RF factor is given by the distance travelled by the solute divided by distance traveled by solvent .…
Q: While using organic solvent like chloroform (CHCl3) in the visible region measurements what cuvette…
A: Glass cuvettes are typically used in the wavelength range of visible light, where as quartz tends to…
Q: Can you TYPE the definitions of the all of words please Chromatography TLC Stationary phase Mobile…
A: Most of the time, the compounds we encounter are not in their purest form. They are essentially a…
Q: A retention time of 85 s is obtained from a gas-chromatographic peak. The base width from the…
A:
Q: Calculate the Rf value of a spot if it travelled 1.5 cm while the solvent travelled 2.5 cm. Express…
A: Given : 1. Spot distance = 1.5 cm 2. Solvent distance = 2.5 cm
Q: Calculate the RF value for a spot in a TLC experiment if the solvent move 13.5 cm and the spot move…
A:
Q: How is the retention factor calculated for a specific spot when doing the TLC analysis? O Distance…
A: We have to tell the process of calculating of retention factor for a particular spot. The definition…
Q: Comment on the differences and similarities between a traditional UV light spectrophotometer and the…
A: Answer - UV-Vis spectrometers - UV-Vis spectrometers use a deuterium lamp for the UV that produces…
Q: 5. Using the diagram answer the questions below: Increasing time Sample appled to top of column…
A: To answer question related to chromatography:
Q: What error is introduced into the determination of an Rf value if the top is left off the developing…
A: In chromatographic technique, the components of a mixture are separated by using a developing…
Q: A gel filtration column has a radius (r) of 0.80 cm and a length (l) of 20.0 cm. Calculate the total…
A:
Q: the efficiency of a chromatographic column improves the measure that a.increases the height of the…
A: To determine what improves the efficiency of a chromatographic column:
Q: calculate Rfs for each spot on your chromatogram
A: Retention factor is equal to the ratio of distance travelled by solute divided by the distance…
Q: The peak parameters for your internal standard, evaluated using a capillary column with a length of…
A: HETP is defined as unit of coloumn length sufficient to bring solute in the mobile phase. It is a…
Q: At the completion of the experiment, a student measures the solvent front to be 55 mm from the…
A:
Q: From the experiment in Question 1, the following data were obtained: 2. Distance traveled by spot O…
A:
Q: what will produce a smaller Rf value if the solvent and paper are both changed?
A: Increasing the polarity of solvent will result in increase in Rf value as the compound in even for…
Q: Calculate the Rf value for a spot in a TLC experiment if the solvent moved 13.5 cm and the spot…
A:
Q: In thin layer chromatography, changing the mobile phase will alter the Rf value of a compound. O…
A: This can be solved as follows
Q: Question 19 In AFM, what is the consequence of low scanning time? limited magnification thermal…
A: AFM refers to Atomic Force Microscopy or Scanning force Microscopy. It is a high resolution type of…
Q: What is the Rf of the compound if the compound distance is 2.59 cm and the solvent front distance is…
A: Given that, Distance travelled by solute= 2.59 cm Distance travelled by solvent= 5.81 cm Then ,…
Q: Question 1. Two components in an HPLC separation have retention times that differ by 22 s. The…
A: HPLC ( High performance liquid Chromatography) It is a useful method for separation of substances…
Q: Does the amount of radiation (cpm) increase or decrease as the distance between the sorce and the…
A: Amount of radiation between the source and sensor is determine using the inverse square formula:…
Q: Calculate the Rf value of a spot that travels 5.7 cm, with a solvent front that travels 13 cm.
A: Rf is a retardation factor that is defined as the distance traveled by a substance divided by the…
Q: Rf of spot A in the TLC plate below is: Solvent front A 10 cm OB 8 cm 6 cm 2 cm Origin Select one: O…
A: The Rf value of a component depends on the distance travelled by the component and the distance…
Q: how does TLC determine if a sample is pure?
A: checking for purity of a sample, the known sample which is pure(having only one spot) and has the…

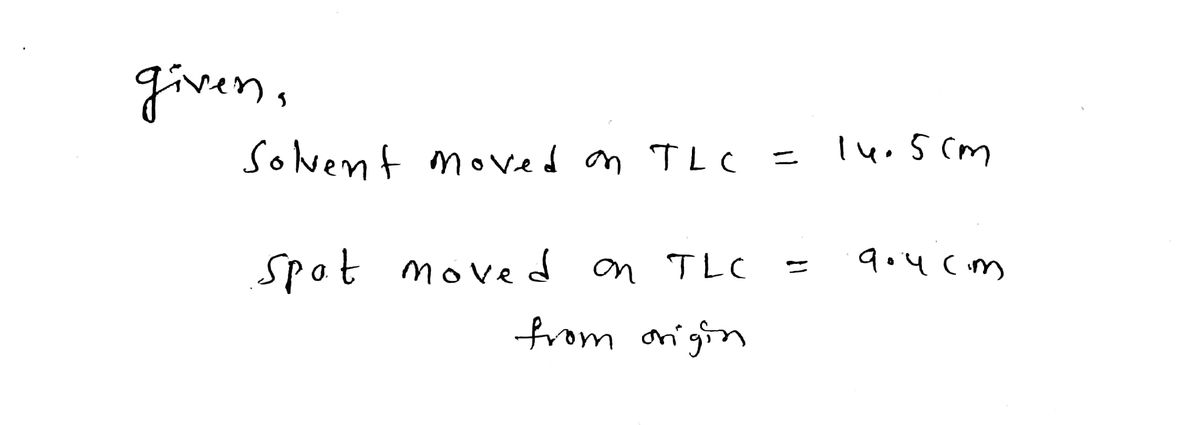
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Calculate the Rf value for a spot in a TLC experiment if the solvent moved 14.5 cm and the spot moved 9.4 cm from the origin. Type answer: 1.54Calculate the Rr value for a spot in a TLC experiment if the solvent moved 14.5 cm and the spot moved 6.1 cm from the origin. Type answer:Calculate the Rf value for a spot in a TLC experiment if the solvent moved 14.9 cm and the spot moved 9.5 cm from the origin.
- Label the graph with the correct axes for a standard curve used in a spectrophotometry experiment. y X Answer Bank Concentration (M) % Transmittance Sample # Absorbance Density (g/L)fill in the column thats empty by finding the standard deviation for each average absorbance, with work please.What is the Rf value given the following information: Height of Chromatography Paper: 5.0 cm Distance from Bottom of Paper to Origin: 1.5 cm Distance from Origin to Spot: 1.6 cm Distance from Origin to Solvent Front: 2.9 cm Your Answer: Answer
- Determine the slope of the linear regression line for a standard curve of concentration versus absorbance using the following data. Concentration (M) Absorbance 2.00x10-5 0.0500 5.00x10-5 0.130 1.00x10-4 0.200 2.00x10-4 0.450 5.00x10-4 1.25 7.50x10-4 1.40 Group of answer choice 5.00x10-4 1.00x10-5 1990 0.0420 0.456 0.296Calculate the RF value for a spot in a TLC experiment if the solvent move 13.5 cm and the spot move 9.1 cm from the originIf the peak width w of both Peak 1 and Peak 2 fro eon 1g is 0.5 minutes, what is the resolution factor between the two peaks? 1.13 Selectivity is Peak 1 (6.0 min, Peak 2 w=0.5 (6.8 min, w=0.5 25 min) min) 20 15 Hold up peak (1.8 min) 10 0. 0. 4. 8. time (minutes) 2. 5. Signal



