
Essential Organic Chemistry, Global Edition
3rd Edition
ISBN: 9781292089034
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 9.9, Problem 23P
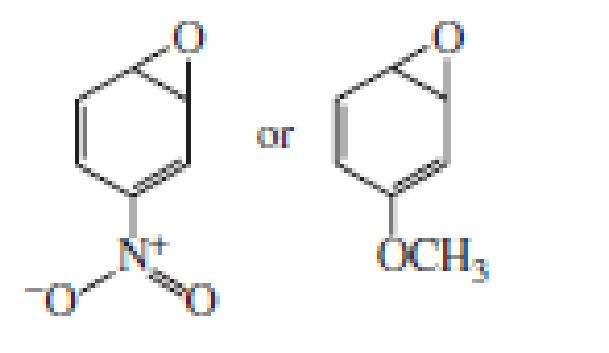
Explain why the two arene oxides in Problem 22 open in opposite directions.
Which compound is more likely to be carcinogenic?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
What kind of crystalline solid is Alumna (Al2O3)?
At night, almost all NOx is present as NO2. (Ie the NO/NOx concentration is very small). Explain why
What is the oxidation number of atom X in H2XO3?
Chapter 9 Solutions
Essential Organic Chemistry, Global Edition
Ch. 9.1 - Draw the structures of straight-chain alcohols...Ch. 9.1 - Prob. 2PCh. 9.1 - Prob. 3PCh. 9.2 - Why are NH3 and CH3NH2 no longer nucleophiles when...Ch. 9.2 - Prob. 5PCh. 9.2 - The observed relative reactivities of primary,...Ch. 9.4 - Which of the following alcohols would dehydrate...Ch. 9.4 - Prob. 10PCh. 9.4 - Prob. 11PCh. 9.4 - Prob. 12P
Ch. 9.4 - Prob. 13PCh. 9.5 - What product will be obtained from the reaction of...Ch. 9.5 - Prob. 15PCh. 9.6 - a. What is each ethers systematic name? 1....Ch. 9.8 - Draw the structure of the following: a....Ch. 9.8 - Prob. 20PCh. 9.8 - Would you expect the reactivity of a five-membered...Ch. 9.9 - Explain why the two arene oxides in Problem 22...Ch. 9.9 - Which compound is more likely to be...Ch. 9.11 - The following three nitrogen mustards were studied...Ch. 9 - What are the common and systematic names of the...Ch. 9 - Prob. 28PCh. 9 - Prob. 29PCh. 9 - Prob. 30PCh. 9 - Prob. 31PCh. 9 - What is the major product obtained from the...Ch. 9 - Draw structures for the following: a....Ch. 9 - Prob. 34PCh. 9 - Prob. 35PCh. 9 - Prob. 36PCh. 9 - Prob. 37PCh. 9 - Ethylene oxide reacts readily with HO.because of...Ch. 9 - Propose a mechanism for each of the following...Ch. 9 - Which of the following ethers would be obtained in...Ch. 9 - Show how each of the following syntheses could be...Ch. 9 - Prob. 42PCh. 9 - Prob. 43PCh. 9 - Prob. 44PCh. 9 - Propose a mechanism for each of the following...Ch. 9 - a. Propose a mechanism for the following reaction:...Ch. 9 - Three arene oxides can be obtained from...Ch. 9 - Prob. 48PCh. 9 - The following reaction takes place several times...Ch. 9 - Show how each of the following compounds could be...Ch. 9 - Propose a mechanism for the following reaction:Ch. 9 - Propose a mechanism for the following reaction:Ch. 9 - What alkenes would you expect to be obtained from...Ch. 9 - Triethylenemelamine (TEM) is an antitumor agent....Ch. 9 - When a diol that has OH groups on adjacent carbons...Ch. 9 - What product is obtained when...Ch. 9 - Prob. 58P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Give the balanced chemical equation when reacting C5H12O with H3O+Cl-arrow_forwardWhat are lanthanoids and actinoids?arrow_forwardHydrogen peroxide is not only an antiseptic but also an oxidizing agent. The following equation shows the reaction of hydrogen peroxide with acetaldehyde to give acetic acid: C2H4O(l) + H2O2(l) → C2H4O2(l) + H2O(l) In this reaction, which species is oxidized and which species is reduced? Which species is the oxidizing agent and which species is the reducing agent?arrow_forward
- The complete oxidation of glucose during cellular respiration is given by the equation below. The free energy change (AG) for the reaction is -2880 kJ/mol and the entropy change (AS) is 0.259 kJ x K-¹ x mol-¹ at 298 K. Calculate the enthalpy change (AH) for the reaction. C6H12O6 + 6 O2 → 6 CO₂ + 6H₂O Group of answer choices 746 kJ/mol 2957 kJ/mol -2803 kJ/mol 2803 kJ/molarrow_forwardWhat is the co-product of the reaction? How is it formed? What is the polarity of the co-product?arrow_forwardWhat is the balanced molecular equation for the complete combustion (in the presence of excess oxygen) of: C21H44 (a component of paraffin wax used in candles)?arrow_forward
- Given the following Reaction: 2N2202 (s) + 2H2O (1) ® 4NaOH (s) + O2 (8) DH°= -126 kJ Explain whether this is an endothermic or exothermic reaction. Provide details in your answer.arrow_forwardWhat is the sum of all the coeifficients of the reactants and products of the balanced following equation?C8H18O3(l) + O2(g) → H2O(g) + CO2(g) 15 30 13 29 22arrow_forwardWhat is the balanced chemical equation for each of the following compounds and calculate the Chemical Oxygen Demand (COD) for each. Toulene - C7H8 Propionic Acid - C3H6O2 Benzene - C6H6arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole
Liquids: Crash Course Chemistry #26; Author: Crash Course;https://www.youtube.com/watch?v=BqQJPCdmIp8;License: Standard YouTube License, CC-BY
Chemistry of Group 16 elements; Author: Ch-11 Chemical Engg, Chemistry and others;https://www.youtube.com/watch?v=5B1F0aDgL6s;License: Standard YouTube License, CC-BY