
Interpretation:
The stereoisomers formed in the reaction of acid catalysed dehydration of 3,4-dimethyl-3-hexanol and the major product has to be determined.
Concept Introduction:
Dehydration reaction:
Removal of water molecule from the reaction when the alcohol is treated with strong acid like sulfuric acid is known as dehydration reaction, for example
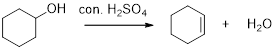
Alcohols are reacts with acids like hydrochloric acid or hydrobromic acid to yield the corresponding carbocation intermediates and then the carbocation intermediate undergoes elimination reaction to give a corresponding
The stability of carbocation is given below,
Tertiary carbocation is more stable than the secondary and primary.
Cis–trans isomerism (or) geometric isomerism or configurational isomerism:
- If the
functional groups are in the same side of the carbon chain, then it is called cis isomer. - If the functional groups are in opposite to each other in the carbon chain, then it is called Trans isomer.
- If two similar functional groups are in same side which is called as Z-isomer.
- If two similar functional groups are opposite side which is called as E-isomer.
Want to see the full answer?
Check out a sample textbook solution
Chapter 9 Solutions
Essential Organic Chemistry, Global Edition
- Acid-catalyzed dehydration of 3-methyl-2-pentanol gives three alkenes: 3-methyl-1-pentene, 3-methyl-2-pentene, and 3-methylenepentane. Draw the structure of the carbocation intermediate leading to the formation of 3-methyl-2-pentene.arrow_forwardWhich is an alkylation product of benzene? NO2 CH3 NH2 В А A Вarrow_forwardName the following compound. CI Br O2-bromo-4-chloro-6-oxyoxane ○ 6-bromo-4-chloro-2-oxyoxane O2-bromo-4-chlorocyclohexanoate ○ 5-bromo-3-chloropentanoic acid lactone ○ 1-bromo-3-chloropentanoic acid lactonearrow_forward
- Select the best method for carrying out the following synthesis: .COOH Br CH, 1. H,C-c-a/AICI, CH3 1. Br /FeBry 1. CH;CI/AICI, 1. KMNO, H, A 1. CH,CI/ AICI, 2. CH,CI/AICI, 2. Bry /FeBr 2. CH,CI/AICI, 2. KMNO H*, A 2. KMNO, H*, A 3. KMNO H", A 3 KMNO, H".A 3. Br, /FeBr, 3. Br/FeBr3 3. Br/FeBr, II III IV O IlI only O V only O Vonly O Il only O l onlyarrow_forwardIf 1-bromopentane is heated in acetone containing NaOH, what is the alkane produced? Draw and explain the step-by-step mechanism of the production of the compoundarrow_forwardProvide the major organic product of the following reaction. but-1-yne 1) Sia₂BH 2) H₂O₂, OH What is it called when an enol converts to a keto? A) Keto-enol isomerism B) Keto-enol tater tot-ism C) Keto-enol conversion D) Keto-enol tautomerismarrow_forward
- The southern pine beetle utilizes a multi-component aggregation pheromone (one component shown below) to start mass colonization of healthy trees. The biosynthetic pathway involves the cyclization of this acetal from the straight chain structure. Draw the straight chain structure that could be used to form this acetal. Use wedges and dashes to correctly depict the stereochemistry. HgC O OH aarrow_forwardWhich of the compound(s) is expected to be the major product in the acid-catalyzed elimination of 2-Methylcyclohexanol? O 4-Methylcyclohexene O 1-Methylcyclohexene O 3-Methylcyclohexene O 2-Methylcyclohexene O Methylenecyclohexanearrow_forwardPredict the b-elimination product(s) formed when each bromoalkane is treated with sodium ethoxide in ethanol. If two or more products might be formed, predict which is the major productarrow_forward
- Which of the following sequences of reactions would convert toluene to 2-bromo-4-cyanotoluene? ? Br CN Nitration, bromination, reduction, diazotization, reaction with cyanide anion Bromination, nitration, reduction, diazotization, reaction with cyanide anion Bromination, nitration, diazotization, reduction , reaction with cyanide anion Nitration, bromination, diazotization, reduction, reaction with cyanide anionarrow_forwardRank the following alcohols in order of increasing ease of acid-catalyzed dehydration. Provide the structure of the dehydration product (alkene) from each alcohol. OH OH 3 1 a OHarrow_forwardexplain why cis-2-chloro-1-cyclohexanol yields a cyclohexanone but the trans isomer yields cyclohexene oxidearrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

