
AVC LOOSELEAF CHEMISTRY W/CONNECT 2 SEM
13th Edition
ISBN: 9781260987164
Author: Chang
Publisher: MCG CUSTOM
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9.6, Problem 3RCF
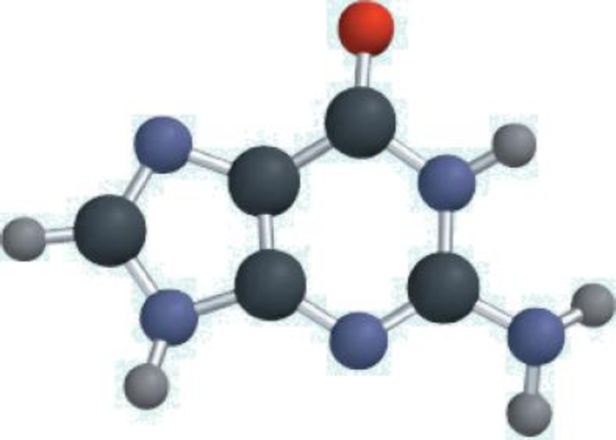
The molecular model shown here represents guanine, a component of a DNA molecule. Only the connections between the atoms are shown in this model. Draw a complete Lewis structure of the molecule, showing all the multiple bonds and lone pairs. (For color code, click here.)
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
<
Complete the following structural formula for a neutral molecule by adding H atoms to complete the valence of each
atom. Do not introduce any double or triple bonds.
Then complete the Lewis diagram by adding any unshared electron pairs needed, so that each atom except H has a
complete octet.
[Review Topics]
[References]
Use the References to access important values if needed for this question.
Br Br
C—C—
Write the molecular formula in the order CHX, where X stands for Cl or Br.
Submit Answer
The number of unshared pairs in the Lewis diagram
unshared pair(s).
Retry Entire Group 9 more group attempts remaining
Previous
Email Instructor
Next
Save and E
The compound XeCl2F2 can exist in two different forms. One form is polar and the other form is non-polar. Draw a valid Lewis structure of XeCl2F2. Then, draw two 3-dimensional representations of this molecule (including all lone pairs); one that shows the polar form with a correct dipole arrow and the other that shows the non-polar form of the molecule.
3. Carbon atoms will bond with each other to form a chain, often with the peripheral atoms being hydrogen. These compounds, hydrocarbons, are organic compounds composed only of carbon and hydrogen. However, when hydrogen atoms are substituted with other elements, such as fluorine, this changes the properties of the molecule. Methylfluoride (CH3F), a substituted hydrocarbon, is used in semiconductor processing. Figure 1 depicts an incorrect Lewis structure for CH3F.
(QUestion shown in photo)
Which reason best explains why this structure is incorrect?
a) Hydrogen lacks an octet.
b) Too few electrons are used.
c) Carbon cannot exceed an octet.
d) Fluorine atom has too many electrons.
Chapter 9 Solutions
AVC LOOSELEAF CHEMISTRY W/CONNECT 2 SEM
Ch. 9.1 - Prob. 1RCFCh. 9.1 - Using only the periodic table, determine the Lewis...Ch. 9.1 - To what group on the periodic table does element X...Ch. 9.2 - Use Lewis dot symbols to represent the formation...Ch. 9.2 - Use Lewis dot symbols to represent the formation...Ch. 9.3 - Which of the following compounds has a larger...Ch. 9.3 - Arrange the compounds NaF, MgO, and AIN in order...Ch. 9.4 - Why is it not possible for hydrogen to form double...Ch. 9.4 - Arrange the bonds NN, NN, and NN in order of...Ch. 9.5 - Which of the following bonds is covalent, which is...
Ch. 9.5 - Rank the bonds BH, CCl, and PP in order of...Ch. 9.5 - Classify the following bonds as covalent, polar...Ch. 9.5 - Identify the electrostatic potential maps shown...Ch. 9.6 - Write the Lewis structure for carbon disulfide...Ch. 9.6 - Write the Lewis structure for formic acid (HCOOH).Ch. 9.6 - Write the Lewis structure for the nitrite ion...Ch. 9.6 - Write the Lewis structure for PCl3.Ch. 9.6 - Write the Lewis structure for CN.Ch. 9.6 - The molecular model shown here represents guanine,...Ch. 9.7 - Write formal charges for the nitrite ion (NO2).Ch. 9.7 - Draw the most reasonable Lewis structure of a...Ch. 9.7 - Write the formal charges on H, C, and N,...Ch. 9.7 - Write the formal charges on O and Cl,...Ch. 9.7 - Consider three possible atomic arrangements for...Ch. 9.8 - Draw three resonance structures for the...Ch. 9.8 - Draw three resonance structures for SO2.Ch. 9.8 - The molecular model shown here represents...Ch. 9.9 - Prob. 9PECh. 9.9 - Prob. 10PECh. 9.9 - Prob. 11PECh. 9.9 - Write the Lewis structure of sulfur tetrafluoride...Ch. 9.9 - Both boron and aluminum tend to form compounds in...Ch. 9.9 - Draw a Lewis structure for IF5.Ch. 9.9 - Prob. 3RCFCh. 9.10 - Calculate the enthalpy of the reaction...Ch. 9.10 - For the reaction H2(g)+C2H4(g)C2H6(g) (a) Estimate...Ch. 9.10 - Why does Hrxn calculated using bond enthalpies not...Ch. 9.10 - Using bond enthalpy values, predict the enthalpy...Ch. 9 - What is a Lewis dot symbol? To what elements does...Ch. 9 - Use the second member of each group from Group 1A...Ch. 9 - Prob. 9.3QPCh. 9 - Write Lewis dot symbols for the following ions:...Ch. 9 - Write Lewis dot symbols for the following atoms...Ch. 9 - Explain what an ionic bond is.Ch. 9 - Prob. 9.7QPCh. 9 - Name five metals and five nonmetals that are very...Ch. 9 - Name one ionic compound that contains only...Ch. 9 - Name one ionic compound that contains a polyatomic...Ch. 9 - Prob. 9.11QPCh. 9 - In which of the following states would NaCl be...Ch. 9 - Prob. 9.14QPCh. 9 - An ionic bond is formed between a cation A+ and an...Ch. 9 - Prob. 9.16QPCh. 9 - Use Lewis dot symbols to show the transfer of...Ch. 9 - Prob. 9.18QPCh. 9 - For each of the following pairs of elements, state...Ch. 9 - Prob. 9.20QPCh. 9 - What is lattice energy and what role does it play...Ch. 9 - Explain how the lattice energy of an ionic...Ch. 9 - Specify which compound in the following pairs of...Ch. 9 - Compare the stability (in the solid state) of the...Ch. 9 - Use the Born-Haber cycle outlined in Section 9.3...Ch. 9 - Calculate the lattice energy of calcium chloride...Ch. 9 - What is Lewiss contribution to our understanding...Ch. 9 - Use an example to illustrate each of the following...Ch. 9 - What is the difference between a Lewis dot symbol...Ch. 9 - How many lone pairs are on the underlined atoms in...Ch. 9 - Compare single, double, and triple bonds in a...Ch. 9 - Compare the properties of ionic compounds and...Ch. 9 - Define electronegativity, and explain the...Ch. 9 - List the following bonds in order of increasing...Ch. 9 - Arrange the following bonds in order of increasing...Ch. 9 - Four atoms are arbitrarily labeled D, E, F, and G....Ch. 9 - List the following bonds in order of increasing...Ch. 9 - Classify the following bonds as ionic, polar...Ch. 9 - Classify the following bonds as ionic, polar...Ch. 9 - Summarize the essential features of the Lewis...Ch. 9 - The octet rule applies mainly to the second-period...Ch. 9 - Write Lewis structures for the following molecules...Ch. 9 - Write Lewis structures for the following molecules...Ch. 9 - Write Lewis structures for the following...Ch. 9 - Write Lewis structures for the following...Ch. 9 - The following Lewis structures for (a) HCN, (b)...Ch. 9 - The skeletal structure of acetic acid shown here...Ch. 9 - Prob. 9.49QPCh. 9 - Do formal charges represent actual separation of...Ch. 9 - Write Lewis structures for the following ions: (a)...Ch. 9 - Write Lewis structures for the following ions: (a)...Ch. 9 - Define bond length, resonance, and resonance...Ch. 9 - Is it possible to trap a resonance structure of a...Ch. 9 - Write Lewis structures for the following species,...Ch. 9 - Draw three resonance structures for the chlorate...Ch. 9 - Write three resonance structures for hydrazoic...Ch. 9 - Draw two resonance structures for diazomethane,...Ch. 9 - Draw three resonance structures for the molecule...Ch. 9 - Draw three reasonable resonance structures for the...Ch. 9 - Why does the octet rule not hold for many...Ch. 9 - Give three examples of compounds that do not...Ch. 9 - Because fluorine has seven valence electrons...Ch. 9 - What is a coordinate covalent bond? Is it...Ch. 9 - Prob. 9.65QPCh. 9 - In the vapor phase, beryllium chloride consists of...Ch. 9 - Of the noble gases, only Kr, Xe, and Rn are known...Ch. 9 - Write a Lewis structure for SbCl5. Does this...Ch. 9 - Prob. 9.69QPCh. 9 - Write Lewis structures for the reaction...Ch. 9 - What is bond enthalpy? Bond enthalpies of...Ch. 9 - Explain why the bond enthalpy of a molecule is...Ch. 9 - From the following data, calculate the average...Ch. 9 - For the reaction O(g)+O2(g)O3(g)H=107.2kJ/mol...Ch. 9 - The bond enthalpy of F2(g) is 156.9 kJ/mol....Ch. 9 - For the reaction 2C2H6(g)+7O2(g)4CO2(g)+6H2O(g)...Ch. 9 - Prob. 9.77QPCh. 9 - Which of the following are ionic compounds? Which...Ch. 9 - Prob. 9.79QPCh. 9 - Use ionization energy (see Table 8.2) and electron...Ch. 9 - Prob. 9.82QPCh. 9 - Prob. 9.83QPCh. 9 - Write three reasonable resonance structures for...Ch. 9 - Prob. 9.85QPCh. 9 - Give an example of an ion or molecule containing...Ch. 9 - Draw four reasonable resonance structures for the...Ch. 9 - Prob. 9.88QPCh. 9 - Draw reasonable resonance structures for the...Ch. 9 - Are the following statements true or false? (a)...Ch. 9 - Prob. 9.91QPCh. 9 - Using the following information and the fact that...Ch. 9 - Prob. 9.93QPCh. 9 - Which of the following molecules has the shortest...Ch. 9 - Most organic acids can be represented as RCOOH,...Ch. 9 - Which of the following species are isoelectronic:...Ch. 9 - The following species have been detected in...Ch. 9 - The amide ion, NH2, is a Brnsted base. Represent...Ch. 9 - Draw Lewis structures for the following organic...Ch. 9 - The triiodide ion (I3) in which the I atoms are...Ch. 9 - Compare the bond enthalpy of F2 with the energy...Ch. 9 - Methyl isocyanate (CH3NCO) is used to make certain...Ch. 9 - Prob. 9.103QPCh. 9 - Prob. 9.104QPCh. 9 - Prob. 9.105QPCh. 9 - Draw Lewis structures for the following...Ch. 9 - Prob. 9.107QPCh. 9 - Prob. 9.108QPCh. 9 - Draw Lewis structures for the following organic...Ch. 9 - Write Lewis structures for the following four...Ch. 9 - Prob. 9.111QPCh. 9 - Prob. 9.112QPCh. 9 - Write three resonance structures for (a) the...Ch. 9 - (a) From the following data calculate the bond...Ch. 9 - Prob. 9.115QPCh. 9 - What are the other two reasons for choosing (b) in...Ch. 9 - Prob. 9.117QPCh. 9 - The NO bond distance in nitric oxide is 115 pm,...Ch. 9 - Prob. 9.119QPCh. 9 - Prob. 9.120QPCh. 9 - Prob. 9.121QPCh. 9 - Prob. 9.122QPCh. 9 - Draw a Lewis structure for nitrogen pentoxide...Ch. 9 - In the gas phase, aluminum chloride exists as a...Ch. 9 - The hydroxyl radical (OH) plays an important role...Ch. 9 - Experiments show that it takes 1656 kJ/mol to...Ch. 9 - Calculate Hrxn at 25C of the reaction between...Ch. 9 - Calculate Hrxn at 25C of the reaction between...Ch. 9 - Draw three resonance structures of sulfur dioxide...Ch. 9 - Vinyl chloride (C2H3Cl) differs from ethylene...Ch. 9 - Prob. 9.131QPCh. 9 - The American chemist Robert S. Mulliken suggested...Ch. 9 - Prob. 9.133QPCh. 9 - A student in your class claims that magnesium...Ch. 9 - Shown here is a skeletal structure of borazine...Ch. 9 - Calculate the wavelength of light needed to carry...Ch. 9 - Prob. 9.137QPCh. 9 - From the lattice energy of KCl in Table 9.1 and...Ch. 9 - The species H3+ is the simplest polyatomic ion....Ch. 9 - The bond enthalpy of the CN bond in the amide...Ch. 9 - In 1999 an unusual cation containing only nitrogen...Ch. 9 - Nitroglycerin, one of the most commonly used...Ch. 9 - Give a brief description of the medical uses of...Ch. 9 - Prob. 9.144QPCh. 9 - The isolated O2 ion is unstable so it is not...Ch. 9 - When irradiated with light of wavelength 471.7 nm,...Ch. 9 - Recall from Chapter 8 that the product of the...Ch. 9 - The reaction between fluorine (F2) with ethane...Ch. 9 - A new allotrope of oxygen, O4, has been reported....Ch. 9 - Because bond formation is exothermic, when two...Ch. 9 - Estimate Hf for sodium astatide (NaAt) according...
Additional Science Textbook Solutions
Find more solutions based on key concepts
The active ingredient in Tylenol and a host of other over-the-counter pain relievers is acetaminophen (C8H9NO2)...
Chemistry: Atoms First
Practice Problem 1.22 Which of the following alkenes can exist as cis-trans isomers? Write their structures. Bu...
Organic Chemistry
During the early part of the 20th century, sulfanilamide (an antibacterial drug) was only administered by injec...
Elementary Principles of Chemical Processes, Binder Ready Version
The structural formula of 1, 2-dimethylbenzene needs to be drawn. Concept introduction: The ring structures of ...
Chemistry: Matter and Change
Fully developed conditions are known to exist for water flowing through a 25-nim-diameer tube at 0.01 kg/s and ...
Fundamentals of Heat and Mass Transfer
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- a. How many sticks did you need to make the skeleton structure?____________ b. How many sticks are left over? ____________ If your model is to obey the octet rule, each ball must have four sticks in it except for hydrogen atom balls, which need and can only have one. Each atom in an octet rule species is surrounded by four pairs of electrons. c. How many holes remain to be filled? ____________ Fill them with the remaining sticks, which represent nonbonding electron pairs. Draw the complete Lewis structure for NH2Cl using lines for bonds and pairs of dots for nonbonding electrons.arrow_forwardDraw Lewis structures showing all valence electrons for these molecules. (a) C2H6 (b) CS2 (c) HCNarrow_forwardWrite the Lewis structure for nitrosyl fluoride, FNO. Using only a periodic table, identify (a) which is the longer bond. (b) which is the stronger bond. (c) which is the more polar bond.arrow_forward
- Use the Molecule Shape simulator (http://openstaxcollege.org/I/6MolecShape) to build a molecule. Starting with the central atom, click on the double bond to add one double bond. Then add one single bond and one lone pair. Rotate the molecule to observe the complete geometry. Name the electron group geometry and molecular structure and predict the bond angle. Then click the check boxes at the bottom and right of the simulator to check your answers.arrow_forwardThe valence shell of an atom in a legitimate Lewis structure (see Figure 2.3) has what in commonwith the valence shell of a noble gas? (Noble gases are stable elements found in the last column ofthe periodic table, e.g., He, Ne, Ar, etc.)arrow_forwardAmmonium bicarbonate, NH4HCO3, contains both ionic and covalent bonds. Draw the Lewis structure of this compound. Explicitly draw all H atoms. • Draw cations and anions in separate sketchers. Do not use the square brackets tool in your answer. opy astearrow_forward
- [References] Ammonium bicarbonate, NH,HCO3, contains both ionic and covalent bonds. Draw the Lewis structure of this compound. Explicitly draw all H atoms. Draw cations and anions in separate sketchers. Do not use the square brackets tool in your answer. C орy аste Carrow_forwardPart ADraw the Lewis structure for HCCH (ethyne).Draw the molecule by placing atoms on the grid and connecting them with bonds. Include all hydrogen atoms and all lone pairs of electrons. Part BDraw the Lewis structure for CS2 (where C is the central atom).Draw the molecule by placing atoms on the grid and connecting them with bonds. Include all lone pairs of electrons.arrow_forwardDecide whether these proposed Lewis structures are reasonable. proposed Lewis structure Is the proposed Lewis structure reasonable? Yes. No, it has the wrong number of valence electrons. Н— Br The correct number is: U No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:* Yes. No, it has the wrong number of valence electrons. .. The correct number is: O Н — С — С — Н No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:* Yes. No, it has the wrong number of valence electrons. The correct number is: || Н — Н- No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:* :0 : :arrow_forward
- Decide whether these proposed Lewis structures are reasonable. proposed Lewis structure Is the proposed Lewis structure reasonable? Yes. H No, it has the wrong number of valence electrons. The correct number is: Н — N— Н No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:* Yes. No, it has the wrong number of valence electrons. The correct number is: Н — С — С — Н No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:* Yes. No, it has the wrong number of valence electrons. The correct number is: :N-N= N | No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:* * If two or more atoms of the same element don't satisfy the octet rule, just enter the chemical symbol as many times as necessary. For example, if two oxygen atoms don't satisfy the octet rule, enter "O,0". :0 : :0 :arrow_forwardDecide whether these proposed Lewis structures are reasonable. proposed Lewis structure Is the proposed Lewis structure reasonable? Yes. No, it has the wrong number of valence electrons. The correct number is: || N - N dlo No, it has the right number of valence electrons but doesn't satisfy the octet rule. Ar The symbols of the problem atoms are:* Yes. No, it has the wrong number of valence electrons. The correct number is:|| N=N No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:* Yes. No, it has the wrong number of valence electrons. The correct number is: || H - C= N No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:* * If two or more atoms of the same element don't satisfy the octet rule, just enter the chemical symbol as many times as necessary. For example, if two oxygen atoms don't satisfy the octet rule, enter "O,0". Explanation Check ©…arrow_forwardDraw the single most stable Lewis structure for the molecule. Include all lone pairs of electrons. NO, BF3, Draw the molecule by placing atoms on the grid and connecting them with bonds. Include all lone pairs of electrons. Use square brackets to denote the overall charge. ICl2-, Draw the single most stable Lewis structure for the molecule. Show the formal charges of all atoms in the correct structure. Include all lone pairs of electrons. OPBr3 (where P is central atom), XeF4arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemical Principles in the LaboratoryChemistryISBN:9781305264434Author:Emil Slowinski, Wayne C. Wolsey, Robert RossiPublisher:Brooks Cole
Chemical Principles in the LaboratoryChemistryISBN:9781305264434Author:Emil Slowinski, Wayne C. Wolsey, Robert RossiPublisher:Brooks Cole Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Chemical Principles in the Laboratory
Chemistry
ISBN:9781305264434
Author:Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:Brooks Cole

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY