
Concept explainers
(a)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: To derive the structure of the compound from its IUPAC name, one should follow the given three steps. The first step involves the identification of parent name and
Answer to Problem 9.42P
The structure corresponding to the given name is,
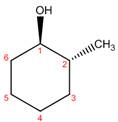
Explanation of Solution
The name of the given compound is

Figure 1
The structure corresponding to the given name is shown in Figure 1.
(b)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: To derive the structure of the compound from its IUPAC name, one should follow the given three steps. The first step involves the identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in either direction. The third step is addition of substituents at appropriate carbon atoms.
Answer to Problem 9.42P
The structure corresponding to the given name is,
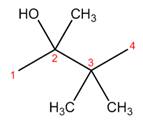
Explanation of Solution
The name of the given compound is
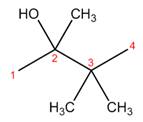
Figure 2
The structure corresponding to the given name is shown in Figure 2.
(c)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: To derive the structure of the compound from its IUPAC name, one should follow the given three steps. The first step involves the identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in either direction. The third step is addition of substituents at appropriate carbon atoms.
Answer to Problem 9.42P
The structure corresponding to the given name is,
Explanation of Solution
The name of the given compound is

Figure 3
The structure corresponding to the given name is shown in Figure 3.
(d)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: To derive the structure of the compound from its IUPAC name, one should follow the given three steps. The first step involves the identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in either direction. The third step is addition of substituents at appropriate carbon atoms.
Answer to Problem 9.42P
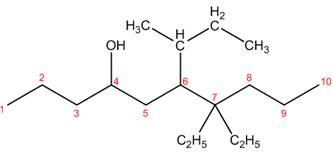
The structure corresponding to the given name is,
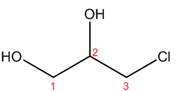
Explanation of Solution
The name of the given compound is
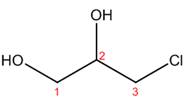
Figure 4
The structure corresponding to the given name is shown in Figure 4.
(e)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: To derive the structure of the compound from its IUPAC name, one should follow the given three steps. The first step involves the identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in either direction. The third step is addition of substituents at appropriate carbon atoms.
Answer to Problem 9.42P
The structure corresponding to the given name is,
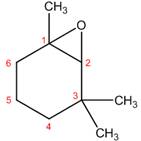
Explanation of Solution
The name of the given compound is

Figure 5
The structure corresponding to the given name is shown in Figure 5.
(f)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: To derive the structure of the compound from its IUPAC name, one should follow the given three steps. The first step involves the identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in either direction. The third step is addition of substituents at appropriate carbon atoms.
Answer to Problem 9.42P
The structure corresponding to the given name is,
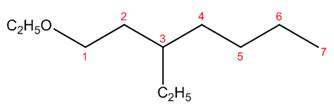
Explanation of Solution
The name of the given compound is

Figure 6
The structure corresponding to the given name is shown in Figure 6.
(g)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: To derive the structure of the compound from its IUPAC name, one should follow the given three steps. The first step involves the identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in either direction. The third step is addition of substituents at appropriate carbon atoms.
The naming of chiral center and geometric isomers are based on Cahn-Ingold-Prelog priority rules. If the priority assigned to each group attached to the chirality center in a molecule is in a clockwise direction, then it is the R-stereoisomer, and if this is counter-clockwise, then it is the S-stereoisomer. R and S-stereoisomer are mirror images of each other.
Answer to Problem 9.42P
The structure corresponding to the given name is,
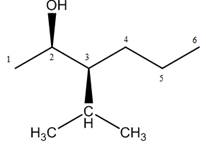
Explanation of Solution
The name of the given compound is

Figure 7
The structure corresponding to the given name is shown in Figure 7.
(h)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: To derive the structure of the compound from its IUPAC name, one should follow the given three steps. The first step involves the identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in either direction. The third step is addition of substituents at appropriate carbon atoms.
The naming of chiral center and geometric isomers are based on Cahn-Ingold-Prelog priority rules. If the priority assigned to each group attached to the chirality center in a molecule is in a clockwise direction, then it is the R-stereoisomer, and if this is counter-clockwise, then it is the S-stereoisomer. R and S-stereoisomer are mirror images of each other.
Answer to Problem 9.42P
The structure corresponding to the given name is,
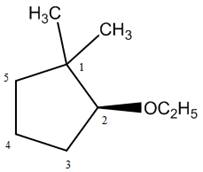
Explanation of Solution
The name of the given compound is

Figure 8
The structure corresponding to the given name is shown in Figure 8.
(i)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: To derive the structure of the compound from its IUPAC name, one should follow the given three steps. The first step involves the identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in either direction. The third step is addition of substituents at appropriate carbon atoms.
Answer to Problem 9.42P
The structure corresponding to the given name is,
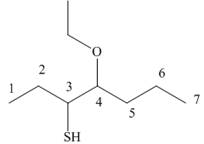
Explanation of Solution
The name of the given compound is
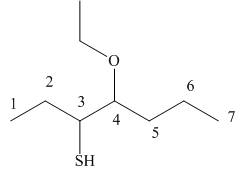
Figure 9
The structure corresponding to the given name is shown in Figure 9.
(j)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: To derive the structure of the compound from its IUPAC name, one should follow the given three steps. The first step involves the identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in either direction. The third step is addition of substituents at appropriate carbon atoms.
Answer to Problem 9.42P
The structure corresponding to the given name is,
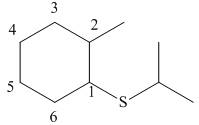
Explanation of Solution
The name of the given compound is

Figure 10
The structure corresponding to the given name is shown in Figure 10.
Want to see more full solutions like this?
Chapter 9 Solutions
Organic Chemistry
- 1. Give the IUPAC name for the following structures. 6-methylhept-4-en-2-one hig a. C. 5-isopropyl-2-heptanone HO pentan-2-yl-5-bromoheptanoate m la Ďarrow_forward64. Which of the following reactant-product relationship is correct? A. 2-methylbutan-2-ol 2-methylbutanoic acid KMnO4 B. 2-methylbutan-2-ol 2-methylbut-3-ene H2504 C. 4-tert-butylcyclohexanol 4- tertbutylcyclohexanone Na2Cr207 D. 4- tertbutylcyclohexanol 4- tertbutylcyclohexanone 7 H2504 65. Choose the correct steps in the dehydration of secondary alcohol; then, arrange in chronological order. 1- Formation of Alkene Il-Formation of Aldehyde III- Formation of Protonated alcohol IV-Undergoes E2 mechanism V-Undergoes E1 mechanism A. V-III-I B. IV-III-II C. III-V-I D. III-IV-II 66. ALL of the following Ether can be prepared by Williamson's method, EXCEPT: A. Ditert-butyl ether B. Ethyl-tert-butyl ether C. anisole D. Both A & B 67. Ethanol is heated with an acidified potassium manganate (VII) solution. What would be the product of this reaction? A. Ethanal B. Ethene C. Ethanoic acid D. Ethanearrow_forwardDraw the products formed when ethylene oxide is treated with each reagent. a. HBr b. H2O(H2SO4) c. [1] CH3CH2O; [2] H2O d. [1] HC ≡ C−; [2] H2O e. [1] −OH; [2] H2O f. [1] CH3S−; [2] H2Oarrow_forward
- Draw the organic product(s) formed when CH3CH2CH2OH is treated with each reagent. a.H2SO4 b.NaH c.HCl + ZnCl2 d.HBr e.SOCl2, pyridine f.PBr3 g.TsCl, pyridine h. [1] NaH; [2] CH3CH2Br [1] i.TsCl, pyridine; [2] NaSH j.POCl3, pyridinearrow_forwardDraw the products formed when p-methylaniline (p-CH3C6H4NH2) is treated with each reagent. a. HCl b. CH3COCl c. (CH3CO)2O d. excess CH3I e. (CH3)2C = O f. CH3COCl, AlCl3 g. CH3CO2H h. NaNO2, HCl i. Part (b), then CH3COCl, AlCl j. CH3CHO, NaBH3CNarrow_forwardGive the structure corresponding to each name. a. 5,6-dimethylhept-2-yne b. 5-tert-butyl-6,6-dimethylnon-3-yne c. (S)-4-chloropent-2-yne d. cis-1-ethynyl-2-methylcyclopentane e. 3,4-dimethylocta-1,5-diyne f. (Z)-6-methyloct-6-en-1-ynearrow_forward
- Does the equilibrium favor the reactants or products in each substitution reaction? a. CH;CH2-NH2 Br CH;CH2-Br + "NH2 b. "CN CN + I-arrow_forwardWhat is the correct name of this compound ? H3C-C CH-CH-CH2-CH3 OH O a. 5-phenoxy-4-hexen-3-ol O b. 2-phenoxy-2-hexen-4-ol O c. 4-hexen-3-ol phenyl ether O d. 4-hydroxy-2-phenoxy-2-hexenearrow_forwardcis-Cyclohexane-1,2-diol can be synthesized from cyclohexene by using which reagent? a.O3 b.OsO4 c.H2SO4 d.mCPBAarrow_forward
- 22. How many of these reactions produce a product named 2-butanol? H₂SO4 H₂O A Br₂ CH₂Cl₂ A. 1 B. 2 C. 3 D. 4 E. 5 1.03 2. Zn/H₂O 1. BH 2. HyOz, NaOH 1. Hg(OAc)2, H₂O 2. NaBH4arrow_forwardWhat is the organic product of the following reaction? ? + 2CH3OH A. C. OH H* E. None of the Above CH3O OCH3 B. D. 10arrow_forwardWhich structures have the correct IUPAC name? CH,OH H OCH, CH, cis-1,2-dimethoxycyclohexane R-2-methoxy-1-propanol II OH H,C HC-OCH, H,C ´OH 2-methoxypropane III trans-1,3-cyclohexanediol IV O A. I, II B. II, IV C. II, II D. I, IVarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





