
Chemistry Atoms First2e
2nd Edition
ISBN: 9781947172647
Author: OpenStax
Publisher: OpenStax College
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9, Problem 78E
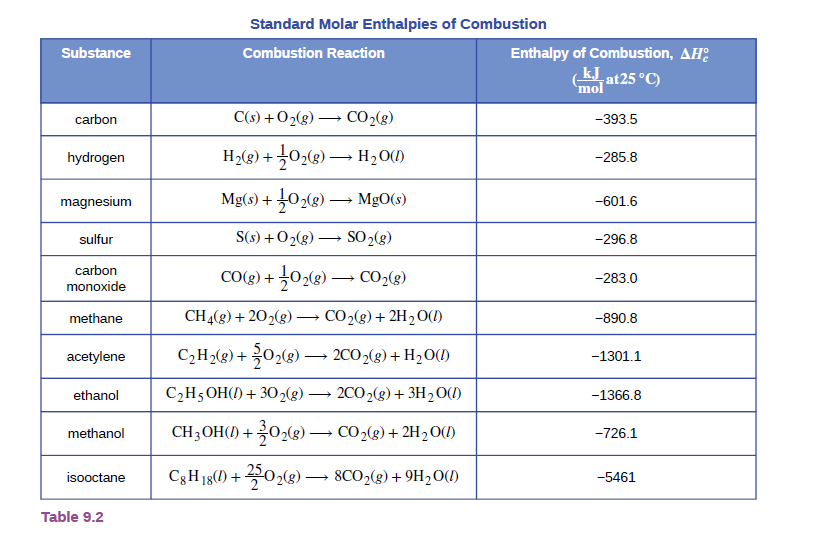
From the data in Table 9.2, determine which of the following fuels produces the greatest amount of heat per gram when burned under standard conditions:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Chapter 9 Solutions
Chemistry Atoms First2e
Ch. 9 - A burning match and a bonfire may have the same...Ch. 9 - Prepare a table identifying several energy...Ch. 9 - Explain the difference between heat capacity and...Ch. 9 - Calculate the heat capacity, in joules and in...Ch. 9 - Calculate the heat capacity, in joules and in...Ch. 9 - How much heat, in joules and in calories, must be...Ch. 9 - How much heat, in joules and in calories, is...Ch. 9 - How much would the temperature of 275 g of water...Ch. 9 - If 14.5 kJ of heat were added to 485 g of liquid...Ch. 9 - A piece of unknown substance weighs 44.7 g and...
Ch. 9 - A piece of unknown solid substance weighs 437.2 g....Ch. 9 - An aluminum kettle weighs 1.05 kg. (a) What is the...Ch. 9 - Most people find waterbeds uncomfortable unless...Ch. 9 - A 500-mL bottle of water at room temperature and a...Ch. 9 - Would the amount of heat measured for the reaction...Ch. 9 - Would the amount of heat absorbed by the...Ch. 9 - Would the amount of heat absorbed by the...Ch. 9 - How many milliliters of water at 23 C with a...Ch. 9 - How much will the temperature of a cup (180 g) of...Ch. 9 - A 45-g aluminum spoon (specific heat 0.88 J/g C)...Ch. 9 - The temperature of the cooling water as it leaves...Ch. 9 - A 70.0-g piece of metal at 80.0 C is placed in 100...Ch. 9 - If a reaction produces 1.506 kJ of heat, which is...Ch. 9 - A 0.500-g sample of KCI is added to 50.0 g of...Ch. 9 - Dissolving 3.0 g of CaCl2(s) in 150.0 g of water...Ch. 9 - When 50.0 g of 0.200 M NaCl(aq) at 24.1 C is added...Ch. 9 - The addition of 3.15 g of Ba(OH)28H2O to a...Ch. 9 - The reaction of 50 mL of acid and 50 mL of base...Ch. 9 - If the 3.21 g of NH4NO3 in Example 9.6 were...Ch. 9 - When 1.0 g of fructose, C6H12O6(s), a sugar...Ch. 9 - When a 0.740-g sample of trinitrotoluene (TNT),...Ch. 9 - One method of generating electricity is by burning...Ch. 9 - The amount of fat recommended for someone with a...Ch. 9 - A teaspoon of the carbohydrate sucrose (common...Ch. 9 - What is the maximum mass of carbohydrate in a 6-oz...Ch. 9 - A pint of premium ice cream can contain 1100...Ch. 9 - A serving of a breakfast cereal contains 3 g of...Ch. 9 - Which is the least expensive source of energy in...Ch. 9 - Explain how the heat measured in Example 9.5...Ch. 9 - Using the data in the check your learning section...Ch. 9 - Calculate the enthalpy of solution( H for the...Ch. 9 - Calculate H for the reaction described by the...Ch. 9 - Calculate the enthalpy of solution ( H for the...Ch. 9 - Although the gas used in an oxyacetylene torch...Ch. 9 - How much heat is produced by burning 4.00 moles of...Ch. 9 - How much heat is produced by combustion of 125 g...Ch. 9 - How many moles of isooctane must be burned to...Ch. 9 - What mass of carbon monoxide must be burned to...Ch. 9 - When 2.50 g of methane burns in oxygen, 125 kJ of...Ch. 9 - How much heat is produced when loo mL of 0.250 M...Ch. 9 - A sample of 0.562 g of carbon is burned in oxygen...Ch. 9 - Before the introduction of chlorofluorocarbons,...Ch. 9 - Homes may be heated by pumping hot water through...Ch. 9 - Which of the enthalpies of combustion in Table 9.2...Ch. 9 - Does the standard enthalpy of formation of H2O(g)...Ch. 9 - Joseph Priestly prepared oxygen in 1774 by heating...Ch. 9 - How many kilojoules of heat will be released when...Ch. 9 - How many kilojoules of heat will be released when...Ch. 9 - The following sequence of reactions occurs in the...Ch. 9 - Both graphite and diamond burn....Ch. 9 - From the molar heats of formation in Appendix G,...Ch. 9 - Which produces more heat?...Ch. 9 - Calculate H for the process Sb(s)+52Cl2(g)SbCl5(s)...Ch. 9 - Calculate H for the process...Ch. 9 - Calculate H for the process Hg2Cl2(s)2Hg(l)+Cl2(g)...Ch. 9 - Calculate H for the process Co3O4(s)3Co(s)+202(g)...Ch. 9 - Calculate the standard molar enthalpy of formation...Ch. 9 - Using the data in Appendix G, calculate the...Ch. 9 - Using the data in Appendix G, calculate the...Ch. 9 - The following reactions can be used to prepare...Ch. 9 - The decomposition of hydrogen peroxide, H2O2, has...Ch. 9 - Calculate the enthalpy of combustion of propane,...Ch. 9 - Calculate the enthalpy of combustion of butane,...Ch. 9 - Both propane and butane are used as gaseous fuels....Ch. 9 - The white pigment TiO2 is prepared by the reaction...Ch. 9 - Water gas, a mixture of H2 and CO2 is an important...Ch. 9 - In the early days of automobiles, illumination at...Ch. 9 - From the data in Table 9.2, determine which of the...Ch. 9 - The enthalpy of combustion of hard coal averages...Ch. 9 - Ethanol, C2H5OH, is used as a fuel for motor...Ch. 9 - Among the substances that react with oxygen and...Ch. 9 - How much heat is produced when 1.25 g of chromium...Ch. 9 - Ethylene, C2H2, a byproduct from the fractional...Ch. 9 - The oxidation of the sugar glucose, C6H12O6, is...Ch. 9 - Propane, C3H8, is a hydrocarbon that is commonly...Ch. 9 - During a recent winter month in Sheboygan,...Ch. 9 - Which bond in each of the following pairs of bonds...Ch. 9 - Using the bond energies in Table 9.3, determine...Ch. 9 - Using the bond energies in Table 9.3, determine...Ch. 9 - Draw a curve that describes the energy of a system...Ch. 9 - Explain why bonds occur at specific average bond...Ch. 9 - When a molecule can form two different structures,...Ch. 9 - How does the bond energy of HCl(g) differ from the...Ch. 9 - Using the standard enthalpy of formation data in...Ch. 9 - Using the standard enthalpy of formation data in...Ch. 9 - Using the standard enthalpy of formation data in...Ch. 9 - Using the standard enthalpy of formation data in...Ch. 9 - Complete the following Lewis structure by adding...Ch. 9 - Use the bond energy to calculate an approximate...Ch. 9 - Use principles of atomic structure to answer each...Ch. 9 - The lattice energy of LiF is 1023 kJ/mol, and the...Ch. 9 - For which of the following substances is the least...Ch. 9 - The reaction of a metal, M, with a halogen, X2,...Ch. 9 - The lattice energy of LiF is 1023 kJ/mol, and the...Ch. 9 - Which compound in each of the following pairs has...Ch. 9 - Which compound in each of the following pairs has...Ch. 9 - Which of the following compounds requires the most...Ch. 9 - Which of the following compounds requires the most...Ch. 9 - The lattice energy of KF is 794 kJ/mol, and the...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What mass of acetylene, C2H2(g), must be burned to produce 3420 kJ of heat, given that its enthalpy of combustion is 1301 kJ/mol? Compare this with the answer to Exercise 5.91 and determine which substance produces more heat per gram.arrow_forwardA 0.470-g sample of magnesium reacts with 200 g dilute HCl in a coffee-cup calorimeter to form MgCl2(aq) and H2(g). The temperature increases by 10.9 C as the magnesium reacts. Assume that the mixture has the same specific heat as water and a mass of 200 g. (a) Calculate the enthalpy change for the reaction. Is the process exothermic or endothermic? (b) Write the chemical equation and evaluate H.arrow_forwardWhen solid iron burns in oxygen gas (at constant pressure) to produce Fe2O3(s), 1651 kJ of heat is released for every 4 mol of iron burned. How much heat is released when 10.3 g Fe2O3(s) is produced (at constant pressure)? What additional information would you need to calculate the heat released to produce this much Fe2O3(s) if you burned iron in ozone gas, O3(g), instead of O2(g)?arrow_forward
- Water gas, a mixture of carbon monoxide and hydrogen, is produced by treating carbon (in the form of coke or coal) with steam at high temperatures. (See Study Question 83.) C(s) + H2O(g) CO(g) + H2(g) Not all of the carbon available is converted to water gas since some is burned to provide the heat for the endothermic reaction of carbon and water. What mass of carbon must be burned (to CO2 gas) to provide the energy to convert 1.00 kg of carbon to water gas?arrow_forwardThe Romans used calcium oxide, CaO, to produce a strong mortar to build stone structures. Calcium oxide was mixed with water to give Ca(OH)2, which reacted slowly with CO2 in the air to give CaCO3. Ca(OH)2(s) + CO2(g) CaCO3(s) + H2O(g) (a) Calculate the standard enthalpy change for this reaction. (b) How much energy is evolved or absorbed as heat if 1.00 kg of Ca(OH)2 reacts with a stoichiometric amount of CO2?arrow_forward9.41 Under what conditions does the enthalpy change equal the heat of a process?arrow_forward
- In a bomb calorimeter, the reaction vessel is surrounded by water that must be added for each experiment. Since the amount of water is not constant from experiment to experiment, the mass of water must be measured in each case. The heat capacity of the calorimeter is broken down into two parts: the water and the calorimeter components. If a calorimeter contains 1.00 kg water and has a total heat capacity of 10.84 kJ/C, what is the heat capacity of the calorimeter components?arrow_forwardThe equation for the combustion of butane, C4H10, is C4H10(g)+132O2(g)4CO2(g)+5H2O(g) Which one of the following generates the least heat? Why? a Burning one mole of butane b Reacting one mole of oxygen with excess butane c Producing one mole of carbon dioxide by burning butane d Producing one mole of water by burning butanearrow_forwardAssume 200. mL of 0.400 M HCl is mixed with 200. mL of 0.400 M NaOH in a coffee-cup calorimeter The temperature of the solutions before mixing was 25.10 C; after mixing and allowing the reaction to occur, the temperature is 27.78 C. What is the enthalpy change when one mole of acid is neutralized? (Assume that the densities of all solutions are 1.00 g/mL and their specific heat capacities are 4.20 J/g K.)arrow_forward
- The enthalpy change for the following reaction is 393.5 kJ. C(s,graphite)+O2(g)CO2(g) (a) Is energy released from or absorbed by the system in this reaction? (b) What quantities of reactants and products are assumed? (c) Predict the enthalpy change observed when 3.00 g carbon burns in an excess of oxygen.arrow_forwardSalicylic acid, C7H6O3, is one of the starting materials in the manufacture of aspirin. When 1.00 g of salicylic acid burns in a bomb calorimeter, the temperature of the bomb and water goes from 23.11C to 28.91C. The calorimeter and water absorb 21.9 kJ of heat. How much heat is given off when one mole of salicylic acid burns?arrow_forwardThe combustion of 1.00 mol liquid methyl alcohol (CH3OH) in excess oxygen is exothermic, giving 727 kJ of heat. (a) Write the thermochemical equation for this reaction. (b) Calculate the enthalpy change that accompanies the burning 10.0 g methanol. (c) Compare this with the amount of heat produced by 10.0 g octane, C8H18, a component of gasoline (see Exercise 5.41).arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
Calorimetry Concept, Examples and Thermochemistry | How to Pass Chemistry; Author: Melissa Maribel;https://www.youtube.com/watch?v=nSh29lUGj00;License: Standard YouTube License, CC-BY