
Chemistry Atoms First2e
2nd Edition
ISBN: 9781947172647
Author: OpenStax
Publisher: OpenStax College
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9, Problem 100E
Use principles of atomic structure to answer each of the following:(4)
(a) The radius of the Ca atom is 197 pm; the radius of the Ca2+ ion is 99 pm. Account for the difference.
(b) The lattice energy of CaO(s ) is -3460 kJ/ mol; the lattice energy of K2O is -2240 kJ/mol. Account for the difference.
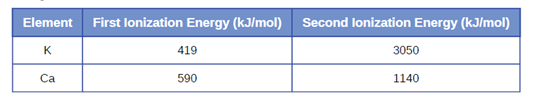
(c) Given these ionization values, explain the difference between Ca and K with regard to their first and second ionization energies.
(d) The first ionization energy of Mg is 738 kJ/mol and that of Al is 578 kj/mol. Account for this difference.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Use principles of atomic structure to answer each of the following:
(1]
(a) The radius of the Ca atom is 197 pm; the radius of the Ca2* ion is 99 pm. Account for the difference.
(b) The lattice energy of CaO(s) is –3460 kJ/mol; the lattice energy of K20 is –2240 kJ/mol. Account for the
difference.
(c) Given these ionization values, explain the difference between Ca and K with regard to their first and second
ionization energies.
Element First lonization Energy (kJ/mol)
Second lonization Energy (kJ/mol)
K
419
3050
Ca
590
1140
(d) The first ionization energy of Mg is 738 kJ/mol and that of Al is 578 kJ/mol. Account for this difference.
Each of the chemically active Period 2 elements forms stable compounds in which it has bonds to fluorine.
(a) What are the names and formulas of these compounds?
(b) Does ∆EN increase or decrease left to right across the period?
(c) Does percent ionic character increase or decrease left to right?
(d) Draw Lewis structures for these compounds
One of the following pictures(Figure 1) represents NaCl and one represents MgO.
Which is which?
(a) is NaCl and (b) is MgO
(b) is NaCl and (a) is MgO
Which has the larger lattice energy?
NaCl
MgO
Chapter 9 Solutions
Chemistry Atoms First2e
Ch. 9 - A burning match and a bonfire may have the same...Ch. 9 - Prepare a table identifying several energy...Ch. 9 - Explain the difference between heat capacity and...Ch. 9 - Calculate the heat capacity, in joules and in...Ch. 9 - Calculate the heat capacity, in joules and in...Ch. 9 - How much heat, in joules and in calories, must be...Ch. 9 - How much heat, in joules and in calories, is...Ch. 9 - How much would the temperature of 275 g of water...Ch. 9 - If 14.5 kJ of heat were added to 485 g of liquid...Ch. 9 - A piece of unknown substance weighs 44.7 g and...
Ch. 9 - A piece of unknown solid substance weighs 437.2 g....Ch. 9 - An aluminum kettle weighs 1.05 kg. (a) What is the...Ch. 9 - Most people find waterbeds uncomfortable unless...Ch. 9 - A 500-mL bottle of water at room temperature and a...Ch. 9 - Would the amount of heat measured for the reaction...Ch. 9 - Would the amount of heat absorbed by the...Ch. 9 - Would the amount of heat absorbed by the...Ch. 9 - How many milliliters of water at 23 C with a...Ch. 9 - How much will the temperature of a cup (180 g) of...Ch. 9 - A 45-g aluminum spoon (specific heat 0.88 J/g C)...Ch. 9 - The temperature of the cooling water as it leaves...Ch. 9 - A 70.0-g piece of metal at 80.0 C is placed in 100...Ch. 9 - If a reaction produces 1.506 kJ of heat, which is...Ch. 9 - A 0.500-g sample of KCI is added to 50.0 g of...Ch. 9 - Dissolving 3.0 g of CaCl2(s) in 150.0 g of water...Ch. 9 - When 50.0 g of 0.200 M NaCl(aq) at 24.1 C is added...Ch. 9 - The addition of 3.15 g of Ba(OH)28H2O to a...Ch. 9 - The reaction of 50 mL of acid and 50 mL of base...Ch. 9 - If the 3.21 g of NH4NO3 in Example 9.6 were...Ch. 9 - When 1.0 g of fructose, C6H12O6(s), a sugar...Ch. 9 - When a 0.740-g sample of trinitrotoluene (TNT),...Ch. 9 - One method of generating electricity is by burning...Ch. 9 - The amount of fat recommended for someone with a...Ch. 9 - A teaspoon of the carbohydrate sucrose (common...Ch. 9 - What is the maximum mass of carbohydrate in a 6-oz...Ch. 9 - A pint of premium ice cream can contain 1100...Ch. 9 - A serving of a breakfast cereal contains 3 g of...Ch. 9 - Which is the least expensive source of energy in...Ch. 9 - Explain how the heat measured in Example 9.5...Ch. 9 - Using the data in the check your learning section...Ch. 9 - Calculate the enthalpy of solution( H for the...Ch. 9 - Calculate H for the reaction described by the...Ch. 9 - Calculate the enthalpy of solution ( H for the...Ch. 9 - Although the gas used in an oxyacetylene torch...Ch. 9 - How much heat is produced by burning 4.00 moles of...Ch. 9 - How much heat is produced by combustion of 125 g...Ch. 9 - How many moles of isooctane must be burned to...Ch. 9 - What mass of carbon monoxide must be burned to...Ch. 9 - When 2.50 g of methane burns in oxygen, 125 kJ of...Ch. 9 - How much heat is produced when loo mL of 0.250 M...Ch. 9 - A sample of 0.562 g of carbon is burned in oxygen...Ch. 9 - Before the introduction of chlorofluorocarbons,...Ch. 9 - Homes may be heated by pumping hot water through...Ch. 9 - Which of the enthalpies of combustion in Table 9.2...Ch. 9 - Does the standard enthalpy of formation of H2O(g)...Ch. 9 - Joseph Priestly prepared oxygen in 1774 by heating...Ch. 9 - How many kilojoules of heat will be released when...Ch. 9 - How many kilojoules of heat will be released when...Ch. 9 - The following sequence of reactions occurs in the...Ch. 9 - Both graphite and diamond burn....Ch. 9 - From the molar heats of formation in Appendix G,...Ch. 9 - Which produces more heat?...Ch. 9 - Calculate H for the process Sb(s)+52Cl2(g)SbCl5(s)...Ch. 9 - Calculate H for the process...Ch. 9 - Calculate H for the process Hg2Cl2(s)2Hg(l)+Cl2(g)...Ch. 9 - Calculate H for the process Co3O4(s)3Co(s)+202(g)...Ch. 9 - Calculate the standard molar enthalpy of formation...Ch. 9 - Using the data in Appendix G, calculate the...Ch. 9 - Using the data in Appendix G, calculate the...Ch. 9 - The following reactions can be used to prepare...Ch. 9 - The decomposition of hydrogen peroxide, H2O2, has...Ch. 9 - Calculate the enthalpy of combustion of propane,...Ch. 9 - Calculate the enthalpy of combustion of butane,...Ch. 9 - Both propane and butane are used as gaseous fuels....Ch. 9 - The white pigment TiO2 is prepared by the reaction...Ch. 9 - Water gas, a mixture of H2 and CO2 is an important...Ch. 9 - In the early days of automobiles, illumination at...Ch. 9 - From the data in Table 9.2, determine which of the...Ch. 9 - The enthalpy of combustion of hard coal averages...Ch. 9 - Ethanol, C2H5OH, is used as a fuel for motor...Ch. 9 - Among the substances that react with oxygen and...Ch. 9 - How much heat is produced when 1.25 g of chromium...Ch. 9 - Ethylene, C2H2, a byproduct from the fractional...Ch. 9 - The oxidation of the sugar glucose, C6H12O6, is...Ch. 9 - Propane, C3H8, is a hydrocarbon that is commonly...Ch. 9 - During a recent winter month in Sheboygan,...Ch. 9 - Which bond in each of the following pairs of bonds...Ch. 9 - Using the bond energies in Table 9.3, determine...Ch. 9 - Using the bond energies in Table 9.3, determine...Ch. 9 - Draw a curve that describes the energy of a system...Ch. 9 - Explain why bonds occur at specific average bond...Ch. 9 - When a molecule can form two different structures,...Ch. 9 - How does the bond energy of HCl(g) differ from the...Ch. 9 - Using the standard enthalpy of formation data in...Ch. 9 - Using the standard enthalpy of formation data in...Ch. 9 - Using the standard enthalpy of formation data in...Ch. 9 - Using the standard enthalpy of formation data in...Ch. 9 - Complete the following Lewis structure by adding...Ch. 9 - Use the bond energy to calculate an approximate...Ch. 9 - Use principles of atomic structure to answer each...Ch. 9 - The lattice energy of LiF is 1023 kJ/mol, and the...Ch. 9 - For which of the following substances is the least...Ch. 9 - The reaction of a metal, M, with a halogen, X2,...Ch. 9 - The lattice energy of LiF is 1023 kJ/mol, and the...Ch. 9 - Which compound in each of the following pairs has...Ch. 9 - Which compound in each of the following pairs has...Ch. 9 - Which of the following compounds requires the most...Ch. 9 - Which of the following compounds requires the most...Ch. 9 - The lattice energy of KF is 794 kJ/mol, and the...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Write electron configurations and Lewis structures for each element. Indicate which of the electrons in the electron configuration are shown in the Lewis structure.(a) Ca(b) Ga(c) As(d) Iarrow_forwardNaCl and KF have the same crystal structure. The only differencebetween the two is the distance that separates cationsand anions. (a) The lattice energies of NaCl and KF are givenin Table 8.1. Based on the lattice energies, would you expectthe Na¬Cl or the K¬F distance to be longer? (b) Use theionic radii given in Figure 7.8 to estimate the Na¬Cl andK¬F distances.arrow_forwardWrite the Lewis dot symbols of the reactants and products in the followingreactions. (First balance the equations.)(a) Sr + Se → SrSe(b) Ca + H2 → CaH2(c) Li + N2 → Li3N(d) Al + S → Al2S3arrow_forward
- Consider the following compounds: BeCl 2 , MgBr 2 , and SrBr 2 . Answer the following questions based on expected periodic trends: (a) Which is expected to have the shortest ionic bonds? (b) Which is expected to have the highest lattice energy? (c) Which is expected to have the lowest melting point?arrow_forward(a) Does the lattice energy of an ionic solid increase or decrease(i) as the charges of the ions increase, (ii) as the sizes of the ionsincrease? (b) Arrange the following substances not listed inTable 8.1 according to their expected lattice energies, listing themfrom lowest lattice energy to the highest: MgS, KI, GaN, LiBr.arrow_forwardWhich set of ionic compounds shows the correct order of increasing lattice energy (lowest-to-highest). (A) NaCl < NaF < KCl < KBr; (B) KBr < KCl < NaCl < NaF; (C) NaF < NaCl < KCl < KBr; (D) KCl < KBr < NaF < NaCl.arrow_forward
- An ionic compound of formula XY2 (X = cation with two positive charges, Y = anion with one negative charge) has the following mass composition: Mg 10.9%, Cl 31.8%, O57.3%. (a) What is the chemical formula and name of the compound? (b) Give the most probable Lewis structure for the ions contained in the compound.arrow_forwardAnswer the following questions that relate to the chemistry of nitrogen. (a) Two nitrogen atoms combine to form a nitrogen molecule, as represented by the following equation. 2 N(g) ® N2(g) Using the table of average bond energies below, determine the enthalpy change, AH, for the reaction. Average Bond Energy (k) mol-1) Bond N-N 160 N=N 420 N°N 950 (b) The reaction between nitrogen and hydrogen to form ammonia is represented below. N2(g) + 3 H2(g)® 2 NH3(g) AH° = -92.2 kJ Predict the sign of the standard entropy change, AS', for the reaction. Justify your answer. (C) The value of AG° for the reaction represented in part (b) is negative at low temperatures but positive at high temperatures. Explain.arrow_forwardCompare the magnitude of the lattice energy for each of the following two ion pairs. (a) Ni²+ and O²- separated by a distance of 394 pm (b) K+ and Br¯ separated by a distance of 358 pmarrow_forward
- Consider the following compounds: CaCl 2 , CaI 2 , and MgCl 2 . Answer the following questions based on expected periodic trends: (a) Which is expected to have t he shortest ionic bonds? (b) Which is expected to have the highest lattice energy? (c) Which is expected to have the lowest melting point?arrow_forwardIodine monochloride and elemental bromine have nearly the same molar mass and liquid density but very different boiling points.(a) What molecular property is primarily responsible for this difference in boiling point? What atomic property gives rise to it? Explain.(b) Which substance has a higher boiling point? Why?arrow_forwardThe first four ionization energies of an element X are 578, 1817, 2745, and 11,577 kJ·mol–1. What is the most likely formula for the most stable ion of Xarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning
Calorimetry Concept, Examples and Thermochemistry | How to Pass Chemistry; Author: Melissa Maribel;https://www.youtube.com/watch?v=nSh29lUGj00;License: Standard YouTube License, CC-BY