
Organic Chemistry - Standalone book
10th Edition
ISBN: 9780073511214
Author: Francis A Carey Dr., Robert M. Giuliano
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9, Problem 32P
Diphenylacetylene can be synthesized by the double dehydrohalogenationof
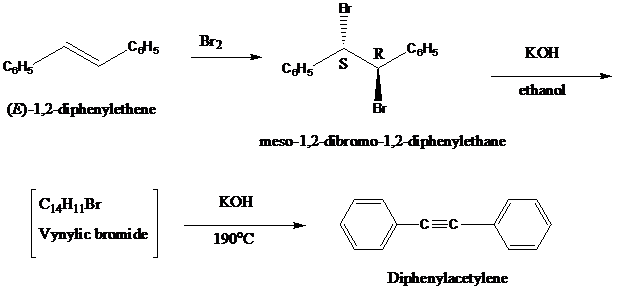
(a) What is the structure, including stereochemistry, of the vinylic bromide?
(b) If the sequence starts with
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Show how the following starting materials are converted to the given product by a series of two pericyclic reactions. Account for the observed stereochemistry.
ASAP
What is the structure of the compounds
• 4-(2-methylprop-1-en-yl)cyclohex-4-ene-1,3-dione
• 3-((methoxymethoxy)methyl)-2,4-dimethylenepentanedioic anhydride
• 2-(phenylthio)phenol
• (Z)-3-acetylhex-2-en-5-ynenitrile
• 6-isopropyl-5,7-di(pentan-3-yl)undecane
Synthesize the following compound starting from benzene.
NO2
Chapter 9 Solutions
Organic Chemistry - Standalone book
Ch. 9.1 - Prob. 1PCh. 9.2 - Prob. 2PCh. 9.4 - How do bond distances and bond strengths change...Ch. 9.5 - Complete each of the following equations to show...Ch. 9.6 - Prob. 5PCh. 9.6 - Which of the alkynes of molecular formula C5H8 can...Ch. 9.7 - Give the structures of three isomeric dibromides...Ch. 9.7 - Prob. 8PCh. 9.9 - Write a series of equations showing how you could...Ch. 9.9 - Write a series of equations showing how to prepare...
Ch. 9.10 - Prob. 11PCh. 9.11 - Give the structure of the enol formed by hydration...Ch. 9.11 - Prob. 13PCh. 9.13 - Prob. 14PCh. 9.14 - Prob. 15PCh. 9 - Provide the IUPAC name for each of the following...Ch. 9 - Prob. 17PCh. 9 - All compounds in Problem 9.17 are isomers except...Ch. 9 - Prob. 19PCh. 9 - Write structural formulas for all the alkynes of...Ch. 9 - Prob. 21PCh. 9 - Prob. 22PCh. 9 - The alkane formed by hydrogenation of...Ch. 9 - Write the structure of the major organic product...Ch. 9 - Write the structure of the major organic product...Ch. 9 - When 2-heptyne was treated with aqueous sulfuric...Ch. 9 - Prob. 27PCh. 9 - Prob. 28PCh. 9 - Prob. 29PCh. 9 - Show by writing appropriate chemical equations how...Ch. 9 - Show by writing appropriate chemical equations how...Ch. 9 - Diphenylacetylene can be synthesized by the double...Ch. 9 - (Z)-9-tricosene [ (Z)-CH3(CH2)7CH=CH(CH2)12CH3 ]...Ch. 9 - Prob. 34PCh. 9 - Prob. 35PCh. 9 - Prob. 36PCh. 9 - Alkynes undergo hydroboration to give...Ch. 9 - Prob. 38DSPCh. 9 - Prob. 39DSPCh. 9 - Prob. 40DSPCh. 9 - Prob. 41DSPCh. 9 - Thinking Mechanistically About Alkynes The...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Please provide the reagents for the following transformations.arrow_forwardShow how the following starting materials are converted to the givenproduct by a series of two pericyclic reactions. Account for the observedstereochemistry.arrow_forwardIn a strongly acidic solution, cyclohexa-1,4-diene tautomerizes to cyclohexa-1,3-diene.Propose a mechanism for this rearrangement, and explain why it is energetically favorablearrow_forward
- When 3-bromo-1-methylcyclohexene undergoes solvolysis in hot ethanol, two productsare formed. Propose a mechanism that accounts for both of these productsarrow_forwardTreatment of 1,2-dibromoethane with the dithiolate dianion shown in the reaction below leads to two products as shown. Draw the structure for each molecular formula and provide a detailed mechanism for the formation of both products.arrow_forwardDraw the structure of an alkene with one double bond that would give the following compound as the only product after ozonolysis followed by H2O2.arrow_forward
- Complete the reaction schemes below providing the reagents required to achieve thetransformation. More than one step may be necessary for each scheme.arrow_forwardProvide the structure of the two organic product(s) which results when 2-bromo-2-methylbutane is treated with sodium ethoxide.arrow_forwardBr Brz CH3 CH3 H3C CH2CI2 H3C Br Electrophilic addition of bromine, Br2; to alkenes yields a 1,2-dibromoalkane. The reaction proceeds through a cyclic intermediate known as a bromonium ion. The reaction occurs in an anhydrous solvent such as CH,Cl). In the second step of the reaction, bromide is the nucleophile and attacks at one of the carbons of the bromonium ion to yield the product. Due to steric clashes, the bromide ion always attacks the carbon from the opposite face of the bromonium ion so that a product with anti stereochemistry is formed. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions Br: :Br: .CH3 H3C H3C CH3 Br:arrow_forward
- The electrophilic addition of HBr to 3-cyclohexylbut-1-ene gives a mixture of two constitutional isomers. These two isomers can also be produced from 3-cylcohexybut-2-ene, but one of which requires different reaction conditions; the electrophilic addition of HBr to 3-cylcohexybut-2-ene produces one of these isomers while the electrophilic addition of HBr to 3-cylcohexybut-2-ene, in the presence of peroxides, produces the other one. Draw the structure for the isomer product that can result from the reactants 3-cyclohexylbut-1-ene and 3-cylcohexybut-2-ene using different reaction conditions. Part 1 of 2 Click and drag to start drawing a structure. C 8 DY 000 8: >arrow_forwardWhat is the major product formed from reacting cyclohexene in aqueous Br2, followed by treatment with potassium tert-butoxide?arrow_forwardPropose a mechanism for the reaction of benzoic acid with oxalyl chloride. This mechanismbegins like the thionyl chloride reaction, to give a reactive mixed anhydride. Nucleophilicacyl substitution by chloride ion gives a tetrahedral intermediate that eliminates a leavinggroup, which then fragments into carbon dioxide, carbon monoxide, and chloride ion.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY