
Concept explainers
Interpretation:The intermolecular forces present in the spilled oil from a pipeline and the pipeline oil have to explain.
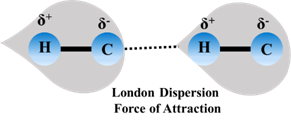
Concept Introduction:In molecules having no permanent dipole like hydrocarbons the electronic atmosphere is supposed to be symmetrical. But this is true only on a time average. Rapid and continuous changes in the intensities of charge concentration in the electron atmosphere may give rise to instantaneous and fluctuating dipoles as shown in the figure. These dipoles may induce further dipoles on adjacent molecules. The resulting weak short range forces are called London Dispersion Forces. The forces increase with increasing polarisibility and hence molar volume among comparable species. Usually these are related to molecular weight but the polarisibility is always important in this force of attraction.
 |
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
Chemistry for Engineering Students
- hat is meant by the term driving forces? Why are mailer spread and energy spread considered to be driving forces?arrow_forwardAre changes in state physical or chemical changes? Explain. What type of forces must be overcome to melt or vaporize a substance (are these forces intramolecular or intermolecular)? Define the molar heat of fusion and molar heat of vaporization. Why is the molar heat of vaporization of water so much larger than its molar heat of fusion? Why does the boiling point of a liquid vary with altitude?arrow_forwardHalothane, which had been used as an anesthetic, has the molecular formula CHBrClCF3. Write the Lewis structure for halothane. Is halothane a polar molecule? Explain your answer. Can hydrogen bonding occur in halothane? Explain.arrow_forward
- Consider the iodine monochloride molecule, ICI. Because chlorine is more electronegative than iodine, this molecule is a dipole. How would you expect iodine monochloride molecules in the gaseous state to orient themselves with respect to each other as the sample is cooled and the molecules begin to aggregate? Sketch the orientation you would expect.arrow_forwardSome ionic compounds contain a mixture of different charged cations. For example, some titanium oxides contain a mixture ofTi2+ and Ti3+ ions. Consider a certain oxide of titanium that is 28.3 1% oxygen by mass and contains a mixture ofTi2+ and Ti3+ ions. Determine the formula of the compound and the relative numbers ofTi2+ and Ti3+ ions.arrow_forwardWhat types of forces exist between the individual particles in an ionic solid? Are these threes relatively strong or relatively weak?arrow_forward
- 88.why does evaporation of water from a surface cause cooling of the surface? evaporation causes the solute molecules left behind to absorb heat. evaporation causes the expansion of water, which helps to extract heat from the surface od and organims evaporation increases the surface tension of liquid water and causes the absorption of heat evaporation causes water to become a better solvent causing polar molecules and ionic compounds to dissolve easily evaporation leaves behind cooler water and prevents overheatingarrow_forwardHydrogen bonding occurs between O oxygen atoms of different water molecules hydrogen and oxygen of the same water molecule a hydrogen on one water molecule and an oxygen on another water molecule O hydrogen atoms of different water moleculesarrow_forwardTwo students plan and carry out an investigation to determine if ionic or covalent bonds have stronger intermolecular forces; they use salt and sugar. They collect the following data: Sugar (covalent) white Salt (ionic) white rough, very small grains rough, small grains soluble soluble melts easily does not melt Which of the following tests will most help them to infer the strength of intermolecular forces between ionic and covalent compounds? Color Texture, grain size Solubility in water Melting point 58°F Clear 99+arrow_forward
- Consider the melting points of the substances below (see figures for two compounds). Explain the trend in melting point using your knowledge of intermolecular forces. (In the structures below, carbon is black, hydrogen is white, and oxygen is red.) Substance Melting Point (°C) Molar Mass (g/mol) Cl2 −102 71 Ethyl formate (CH3CH2OCHO) −80 74 Propionic acid (CH3CH2COOH) −20 74 Br2 −7.2 160arrow_forward4. What is the relationship between a liquid's molecular weight and the strength of intermolecular bonds (assume the liquid is only compared to other liquids that lo similar intermolecular bonds)? Use the molecular structures and weights of ethano and methanol, the types of intra- and inter- molecular bonds they make and the observations you made of ethanol and methanol in this investigation to support your answer. What is different about water's molecular structure that makes water not follow this relationship when compared to ethanol and methanol?arrow_forwardChemistry The boiling point of a group 4A binary hydrogen compound is CH4-162°C. SiH4- 112°C, GeH4-88°C, SnH4-52°C. Explain why the boiling point increases from CH4 to SnH4arrow_forward
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





