
(a)
Interpretation:
The mechanism for the reaction of 2-butyne with one equivalent of bromine should be given.
Concept Introduction:
Addition of halides to
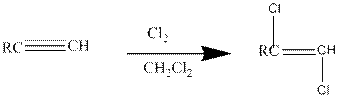
If excess halides are present, a second addition reaction also occurs.
(b)
Interpretation:
The configuration of the product obtained in the above reaction should be identified.
Concept Introduction:
Addition of halides to alkynes: Halogens like chlorine and bromine adds to the alkyne compounds. The general form of the reaction is given below.

If excess halides are present, a second addition reaction also occurs.
Trending nowThis is a popular solution!

Chapter 7 Solutions
Organic Chemistry; Organic Chemistry Study Guide A Format: Kit/package/shrinkwrap
- Select the best reagent expected to convert 3-heptyne to cis-3-heptene. A. NaNH2, NH3 B. Na, NH3 C. H2, Lindlar’s catalyst D. Both A and C E. Both B and Carrow_forward8. Give an example of a carbene and give a reason why CHCl3 readily forms a carbene in NaOH.arrow_forwardWhen free radical halogenation is performed with one of the compounds below, only a single product with the formula C5H11Cl is isolated. Which compound fits this description? Select one: a. 2,2-dimethylpropane b. cyclopentane c. pentane d. 2-methylbutanearrow_forward
- Draw the two major products of this reaction. Use a dash or wedge bond to indicate the stereochemistry of substituents on asymmetric centers. Ignore inorganic byproducts. 1. BHS-THF 2.H2O2, NaOH, H₂O 回arrow_forward2. Draw ALL possible structural and stereochemical (if applicable) isomers. Please include enols (if oxygen is present) and enamines (if nitrogen is present), and any E and Z isomers in this analysis. a. C₂H4O b. a. C₂H₂O C. b. C4H₂O C₂H₂Narrow_forwardOzonolysis (O3 in CH2Cl2) of compound A under reducing conditions (Zn /acetic acid) gives formaldehyde, 2-butanone, and compound B. Catalytic hydrogenation (H2/Pd) of A gives 2,7-dimethylnonane. What is a possible structure for compound A? a. 2,7-Dimethyl-2,8-nonadieneb. 2,7-Dimethyl-1,8-nonadienec. 2,7-Dimethyl-1,6-nonadiened. 2,7-Dimethyl-1,7-nonadienearrow_forward
- Draw the products of the following reactions. If the products can exist as stereoisomers show what stereoisomers are formed. a. cis-2-pentene + Br2/CH2Cl2 b. trans-2-pentene + Br2/CH2Cl2 c. 1- butene + HCl d. methylcyclohexene + HBr e. trans-3-hexene + Br2/CH2Cl2 f. cis-3-hexene + Br2/CH2Cl2 g. 3,3-dimethyl-1-pentene + HBr h. cis-2-butene + HBr i. (Z)-2,3-dichloro-2-butene + H2, Pd/C j. (E)-2,3-dichloro-2-butene + H2, Pd/C k. (Z)-3,4-dimethyl-3-hexene + H2, Pd/C l. (E)-3,4-dimethyl-3-hexene + H2, Pd/Carrow_forwarda. Draw the structures and give the common and systematic names for alkynes with molecular formula C7H12. Ignore stereosiomers. (Hint: There are 14.) b. How many would there be if stereoisomers are included?arrow_forward57. The order of decreasing ease of dehydrohalogenation OCH₂C I. >-CH2CH2CI A. I>II>III>IV B. CH3CH₂CHCICH3 C. II. CHOHCH₂CH3 D. CH3CHBRCH₂CH3 CH₂CH2C1 B. III>II>I>IV 58. Which of the following reactions will produce the highest percentage yield of 1-butene. OH A. CH3CH₂CH(N* (CH3)3)CH3 alc. KOH conc. H₂SO4 heat NaOC₂H5 III. C₂H5OH of the following compounds is: CH2CH₂CI CH₂CH₂C1 CH₂ -CH₂CH2CI C. III>IV>II> I IV. D. IV>III>II>Iarrow_forward
- (R)-2-butanol reacts with phosphorus tribromide to give A (C4H9Br). Treatment of A with sodium cyanide in DMF gives B (C5H9)N. B is optically active. Draw the structure of B. • Use the wedge/hash bond tools to indicate stereochemistry where it exists. Show stereochemistry in a meso compound. Sn [F ? ChemDoodleⓇarrow_forwardDraw the structural formula for the product formed upon hydroboration/oxidation of the Alkene below.arrow_forward6. What is the product of the following reaction? A. B. (C6H5)3P-CH2 C. D.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

