
Pearson eText for Essential Organic Chemistry -- Instant Access (Pearson+)
3rd Edition
ISBN: 9780137533268
Author: Paula Bruice
Publisher: PEARSON+
expand_more
expand_more
format_list_bulleted
Question
Chapter 7.11, Problem 22P
Interpretation Introduction
Interpretation:
The products obtianed in the reaction of 1,3,5-hexatriene with one equivalent
Concept Introduction:
Electrophile: It is positively charged species which seeks for negative charge and hence accepts pair of electrons from negatively charged species (Nucleophiles) which results in the formation of
Reactions of conjugated dienes:
The conjugated diene undergoes addition reaction in presence of electrophile such that the addition only occurs on double bonds in diene.
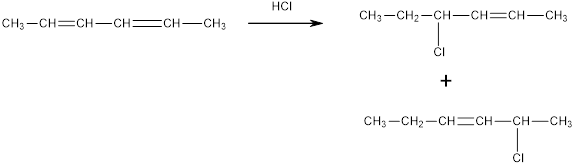
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What three alkenes (excluding stereoisomers) can be used to prepare 3chloro-3-methylhexane by addition of HCl?
1,3-Cyclohexadiene and 1,4-cyclohexadiene can be distinguished by reaction with ozone and then treatment with zinc. Draw the structures of the products produced by ozonolysis of the two compounds.
Two substitution products result from the
reaction between 3-chloro-3-methyl-1-
butene with sodium acetate (CH3COO – Na +)
in acetic acid under SN1.
Identify the products.
Chapter 7 Solutions
Pearson eText for Essential Organic Chemistry -- Instant Access (Pearson+)
Ch. 7.4 - Prob. 1PCh. 7.5 - Prob. 3PCh. 7.6 - a. Predict the relative bond lengths of the three...Ch. 7.6 - Prob. 5PCh. 7.6 - Prob. 6PCh. 7.7 - Prob. 7PCh. 7.7 - Prob. 8PCh. 7.7 - Prob. 9PCh. 7.7 - Prob. 10PCh. 7.8 - Which member of each pair is the stronger acid? a....
Ch. 7.8 - Which member of each pair is the stronger base? a....Ch. 7.9 - Prob. 13PCh. 7.9 - Which species in each of the following pairs is...Ch. 7.9 - Prob. 16PCh. 7.11 - Prob. 18PCh. 7.11 - Prob. 19PCh. 7.11 - Prob. 20PCh. 7.11 - Prob. 21PCh. 7.11 - Prob. 22PCh. 7.12 - Prob. 23PCh. 7.12 - Prob. 24PCh. 7.15 - Prob. 25PCh. 7.15 - Which of the following are aromatic? A...Ch. 7.17 - Prob. 29PCh. 7.17 - Prob. 30PCh. 7 - Prob. 31PCh. 7 - Prob. 32PCh. 7 - Prob. 33PCh. 7 - Prob. 34PCh. 7 - Prob. 35PCh. 7 - Prob. 36PCh. 7 - Which of the compounds in each of the following...Ch. 7 - Prob. 39PCh. 7 - Prob. 40PCh. 7 - Prob. 41PCh. 7 - Prob. 42PCh. 7 - Prob. 43PCh. 7 - Prob. 44PCh. 7 - Prob. 45PCh. 7 - Which species in each of the pairs in Problem 45...Ch. 7 - Rank the following anions in order from most basic...Ch. 7 - a. Which oxygen atom has the greater electron...Ch. 7 - Prob. 49PCh. 7 - Which compound is the strongest base?Ch. 7 - Prob. 51PCh. 7 - Prob. 52PCh. 7 - Prob. 53PCh. 7 - Prob. 54PCh. 7 - Prob. 55PCh. 7 - a. Which dienophile in each pair is more reactive...Ch. 7 - Prob. 57PCh. 7 - Draw the product of each of the following...Ch. 7 - Prob. 59PCh. 7 - Prob. 60PCh. 7 - Prob. 61PCh. 7 - a. In what direction is the dipole moment in...Ch. 7 - Propose a mechanism for each of the following...Ch. 7 - Prob. 1PCh. 7 - Prob. 2PCh. 7 - Prob. 3PCh. 7 - Prob. 4PCh. 7 - Prob. 5PCh. 7 - Prob. 6PCh. 7 - Prob. 7PCh. 7 - Prob. 8PCh. 7 - Prob. 9PCh. 7 - Prob. 10PCh. 7 - Prob. 11PCh. 7 - Prob. 12P
Knowledge Booster
Similar questions
- One compound that contributes to the “seashore smell” at beaches in Hawai‘i is dictyopterene D', a component of a brown edible seaweed called limu lipoa. Hydrogenation of dictyopterene D' with excess H2 in the presence of a Pd catalyst forms butylcycloheptane. Ozonolysis with O3 followed by (CH3)2S forms CH2(CHO)2, HCOCH2CH(CHO)2, and CH3CH2CHO. What are possible structures of dictyopterene D'?arrow_forwardGive the structure of the following compound: 2-(1-chloroethyl)-5-methylheptanalarrow_forwardWhat organic product would you obtain from reaction of 1-pentanol with CrO3, H2O, H2SO4?arrow_forward
- When 2-chloropropane treated with NaOH and 1-chloropropane treated with NaOH separately produce two different functional groups. Provide both reactions and explain the two different functional groups produced.arrow_forwardCompound A is an alkyne that reacts with two equivalents of H2 in the presence of Pd to give 2,4,6-trimethyloctane. Draw the structure of compound A.arrow_forwardTwo products are formed when methylenecyclohexane reacts with NBS? Show how each is formed. Disregard stereoisomers.arrow_forward
- How many allylic substituted bromoalkenes are formed from the reaction of 2-pentene with NBS? Disregard stereoisomers.arrow_forwardA compound with formula C7H12O is treated with sodium borohydride in methanol to yield 2,2-dimethylcylopentanol. Write a reaction scheme showing the structures of the reactant, the reagents, and the product. Will the product be optically active? Explain.arrow_forwardExplain the Preparation of Dihalocarbenes ?arrow_forward
- 1. The ketone 2-heptanone has been identified as contributing to the odor of a number of dairy products, including condensed milk and cheddar cheese. Describe the synthesis of 2-heptanone from acetylene and any necessary organic and inorganic reagents. 2. Compound A has the molecular formula C14H25Br and was obtained by reaction of sodium acetylide (HC≡CNa) )with 1,12-dibromododecane. On treatment of compound A with sodium amide, it was converted to compound B (C14H24). Ozonolysis of compound B gave the diacid HO2C(CH2)12CO2H. Catalytic hydrogenation of compound B over Lindlar palladium gave compound C (C14H26), while hydrogenation over platinum gave compound D (C14H28). Sodium-ammonia reduction of compound B gave compound E (C14H26). Both C and E yielded O═CH(CH2)12CH═O on ozonolysis. Assign structures to compound A through E so as to be consistent with the observed transformations.arrow_forwardAn unknown hydrocarbon A with the formula C6H10 reacts with 1 molar equivalent of H2 over a palladium catalyst to give B C6H12 (Rxn 1). Hydrocarbon A also reacts with OsO4 to give the glycol C (Rxn 2). A gives 5-oxohexanal on ozonolysis (Rxn 3). Draw the structures of A, B, and C. Give the reactions.arrow_forwardFour alkenes are formed from the E1 reaction of 3-bromo-2,3-dimethylpentane and methanol. Draw the structures of the alkenes and rank them according to the amount that would be formed.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY