
(a)
Interpretation:
The diene and dienophile from the given compounds has to be predicted.
Concept Introduction:
Diels-Alder reaction:
A conjugated diene reacts with a compound containing a carbon-carbon double bond. It is a cycloaddition reaction, where two reactants form a cyclic product.
Conformation of Diene:
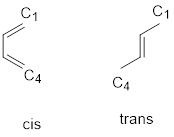
S-cis Conformer: The double bonds are cis about the single bond (s stands for single), in an s-cis conformer.
S-trans Conformer: The double bonds are trans- about the single bond (s stands for single) in an s-trans conformer.
Condition for Diels-Alder reaction:
The conjugate diene must be in an s-cis conformation because when it is in an S-trans conformation, C-1 and C-4 are too far apart to react with the dienophile in a concerted reaction.
(b)
Interpretation:
The diene and dienophile from the given compounds has to be predicted.
Concept Introduction:
Diels-Alder reaction:
A conjugated diene reacts with a compound containing a carbon-carbon double bond. It is a cycloaddition reaction, where two reactants form a cyclic product.
Conformation of Diene:
S-Cis Conformer: The double bonds are cis about the single bond (s stands for single), in an s-cis conformer.
S-trans Conformer: The double bonds are trans- about the single bond (s stands for single) in an s-trans conformer.
Condition for Diels-Alder reaction:
The conjugate diene must be in an s-cis conformation because when it is in an S-trans conformation, C-1 and C-4 are too far apart to react with the dienophile in a concerted reaction.

(c)
Interpretation:
The diene and dienophile from the given compounds have to be predicted.
Concept Introduction:
Diels-Alder reaction:
A conjugated diene reacts with a compound containing a carbon-carbon double bond. It is a cycloaddition reaction, where two reactants form a cyclic product.
Conformation of Diene:
S-Cis Conformer: The double bonds are cis about the single bond (s stands for single), in an s-cis conformer.
S-trans Conformer: The double bonds are trans- about the single bond (s stands for single) in an s-trans conformer.
Condition for Diels-Alder reaction:
The conjugate diene must be in an s-cis conformation because when it is in an S-trans conformation, C-1 and C-4 are too far apart to react with the dienophile in a concerted reaction.

Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
Pearson eText for Essential Organic Chemistry -- Instant Access (Pearson+)
- The following triene undergoes an intramolecular Diels-Alder reaction to give a bicyclic product. Propose a structural formula for the product. Account for the observation that the Diels-Alder reaction given in this problem takes place under milder conditions (at lower temperature) than the analogous Diels-Alder reaction shown in Problem 20.34.arrow_forwardSelect the two compounds that could be used to make Compound V through a Diels-Alder reaction. CN CN Compound V C NC. CN E CN CN H.arrow_forwardHow could the following compounds be synthesized using a Diels–Alder reaction?arrow_forward
- 3) How could you use Diels-Alder reactions to prepare the following products? Show the starting diene and dienophile in each case H a. b. C. H H d. H .CO₂CH3 CNarrow_forward9. For the following Diels-Alder products, propose the correct starting materials needed. D. a. b. C. fo{- ●●● Xox -arrow_forward3. Predict the major product(s) for the following reaction. HCI o°c 4. Predict the product for the following Diels-Alder reaction. 5. Predict the product for the following Diels-Alder reaction. ÇOCH, Aarrow_forward
- A possible side reaction is butadiene reacts with another butadiene via a Diels‐Alder reaction. Draw the structure of that product.arrow_forwardWhat is the major organic product of the following Diels-Alder reaction? Me₂N. A. B. C. D. Me₂N Me₂N Me₂N OHC Me₂N CHO CHO CHO CHO 5 heatarrow_forward3. Show how Diels-Alder reactions might be used to synthesize the following compounds. a. Harrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

