
Concept explainers
(a)
Interpretation: The nucleophile needed to convert
Concept introduction: The replacement or substitution of one
Answer to Problem 7.19P
The nucleophile needed to convert
Explanation of Solution
The reactant of the reaction is
![]()
Figure 1
The reaction of
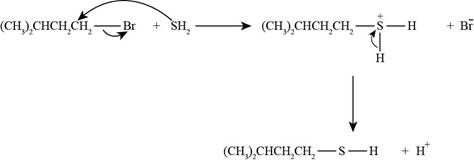
Figure 2
The nucleophile needed to convert
(b)
Interpretation: The nucleophile needed to convert
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron rich chemical species that contains negative charge or lone pair of electrons are known as a nucleophile. In a nucleophilic substitution reaction, nucleophile takes the position of leaving group by attacking the electron deficient carbon atom.
Answer to Problem 7.19P
The nucleophile needed to convert
Explanation of Solution
The reactant of the reaction is
![]()
Figure 3
The reaction of
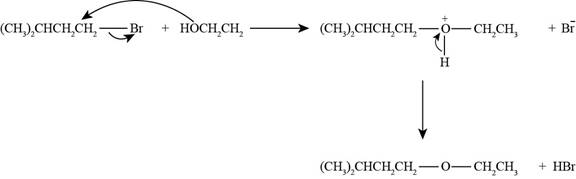
Figure 4
The nucleophile needed to convert
(c)
Interpretation: The nucleophile needed to convert
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron rich chemical species that contains negative charge or lone pair of electrons are known as a nucleophile. In a nucleophilic substitution reaction, nucleophile takes the position of leaving group by attacking the electron deficient carbon atom.
Answer to Problem 7.19P
The nucleophile needed to convert
Explanation of Solution
The reactant of the reaction is
![]()
Figure 5
The reaction of
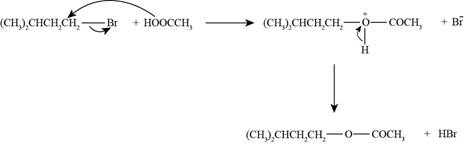
Figure 6
The nucleophile needed to convert
(d)
Interpretation: The nucleophile needed to convert
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron rich chemical species that contains negative charge or lone pair of electrons are known as a nucleophile. In a nucleophilic substitution reaction, nucleophile takes the position of leaving group by attacking the electron deficient carbon atom.
Answer to Problem 7.19P
The nucleophile needed to convert
Explanation of Solution
The reactant of the reaction is
![]()
Figure 7
The nucleophile needed to convert
Want to see more full solutions like this?
Chapter 7 Solutions
Organic Chemistry-Package(Custom)
- 16.41 Draw the products of each reaction. а. HNO3 H2SO4 'NO2 HO b. AICI3 CI С. H. Br2 FeBr3arrow_forward16.69 Use the reactions in this chapter along with those learned in Chapters 11 and 12 to synthesize each compaund. You may use benzene, acetylene (HC=CH), ethanol, ethylene oxide, and any inorganic reagents. a. b. CI он C ON d. NO2arrow_forward16.43 Draw the products of each reaction. a. b. d. HO C. H Lore L sob Br₂ FeBr3 e. NO₂ Br HNO3 H₂SO4 CI AICI 3 Cl₂ FeCl3 SO3 H₂SO4arrow_forward
- 16.41 Draw the products of each reaction. a. b. HO NO₂ mellanrios H HNO3 H₂SO4 Br₂ FeBr3 AICI3 d. e. Br- edmun adtonie F prier f. F (X) napoln NO₂ Cl₂ FeCl3 SO3 H₂SO4 onho sur + Na Sarrow_forward4a. Which of the following is the major product of the following elimination? e OMe CI CI A. Ans: Ans: 4b. Which of the following is the major product of the following elimination? B. A. C. B. C.arrow_forward8) Identify the nucleophile and electrophile in each reaction. А. H. O.. CI CI B.arrow_forward
- Draw the products of radical chlorination and bromination of each compound. For which compounds is a single constitutional isomer formed for both reactions? What must be true about the structure of a reactant for both reactions to form a single product? a. b. C. d. е.arrow_forwardProblem 12.27 Draw the products of each reaction sequence. a. b. |||||| I [2] [2] [1] Δ [1] Aarrow_forward2. Fill in the necessary reagents for each reaction. HO 1. A. В. 2.arrow_forward
- Problem 13.34 Synthesize each compound from cyclohexanol, ethanol, and any other needed reagents. a. b. C. d. e. OH XOH Br OHarrow_forward12. What alkyl halide and nucleophile are needed to prepare the following compounds? a. b. OHarrow_forwardWhich pair of reactants for a Grignard reaction does not give 2-phenylbutan-2-ol after an aqueous workup? a. b. C. 01 O 8 -C-OCH₂CH3 + CH₂MgBr CH3CCH₂CH3 + -MgBr -C-CH3 + CH3CH₂MgBr -C-CH₂CH3 + CH3MgBrarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





