
Concept explainers
(a)
Interpretation:
The configuration of the chiral center has to be identified for given substrate.
Concept Introduction:
Chirality: It refers to a Carbon atom in a molecule that contains four different substituents.
Enantiomers: they are chiral molecules whose mirror images are not superimposable.
R and S nomenclature: it is used to assign the molecule with chiral carbon using CIP rules.
The CIP rules are as follows:
- 1. Select the chiral carbon and assign the numbers to the atoms in the decreasing order of their
atomic mass by placing the hydrogen atom below the plane. - 2. If the numbering follows clockwise direction then the molecule is termed as R and if it follows anti-clockwise direction then molecule is termed as S.
- 3. Suppose if the hydrogen is placed above the plane then the corresponding isomer is just opposite to the resulting isomer.
(b)
Interpretation:
The reaction mechanism and the configurations of substrate and product for the given reaction should be assigned.
Concept Introduction:
Chirality: It refers to a Carbon atom in a molecule that contains four different substituents.
Enantiomers: they are chiral molecules whose mirror images are not superimposable.
R and S nomenclature: it is used to assign the molecule with chiral carbon using CIP rules.
The CIP rules are as follows:
- 1. Select the chiral carbon and assign the numbers to the atoms in the decreasing order of their atomic mass by placing the hydrogen atom below the plane.
- 2. If the numbering follows clockwise direction then the molecule is termed as R and if it follows anti-clockwise direction then molecule is termed as S.
- 3. Suppose if the hydrogen is placed above the plane then the corresponding isomer is just opposite to the resulting isomer.
(c)
Interpretation:
The reaction mechanism and the configurations of substrate and product for the given reaction should be assigned.
Concept Introduction:
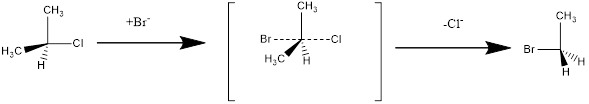
Structure of the substrate plays major role in
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
ORGANIC CHEMISTRYPKGDRL+MLCRL MDL
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





