
Concept explainers
(a)
Interpretation:
The graph of the given data to form an oxygen-binding curve is to be plotted. The oxygen partial pressure at which the given hemoglobin is half-saturated is to be stated. Whether oxygen binding seems to be cooperative or not on the basis of the curve is to be stated.
Concept introduction:
Proteins are the
Answer to Problem 16P
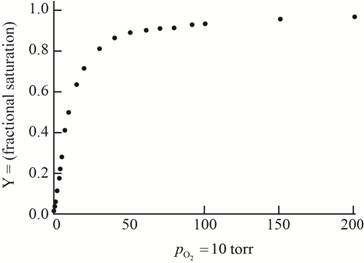
The plot of the given data to form an oxygen-binding curve is shown below.
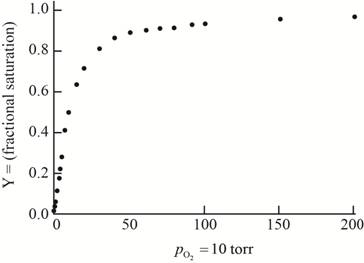
The oxygen partial pressure at which the given hemoglobin is half-saturated is
Explanation of Solution
On the basis of the given data for oxygen-binding for lamprey hemoglobin, the graph of fractional saturation versus partial pressure of oxygen is plotted as follows:
At the oxygen partial pressure,
Thus, this plot between partial pressure of oxygen
(b)
Interpretation:
A hill plot by using the given data is to be stated. Whether the hill plot shows any evidence for cooperativity or not is to be stated. The information about the hill coefficient is to be explained.
Concept introduction:
Proteins are the biomolecules which are composed of the long chain of amino acid residues. The protein which contains oxygen and is present in the red blood cells in the body is known as hemoglobin. It contains iron as well.
The hill equation indicates the two close equations which show the binding of a ligand with the macromolecule.
Answer to Problem 16P
A hill plot by using the given data is shown as,
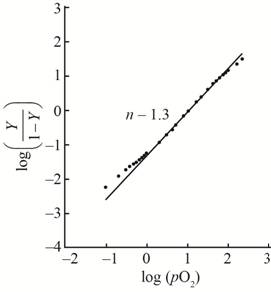
This plot show little bit cooperativity in the center of the curve and its hill coefficient is
Explanation of Solution
The equation that is used to show the hill equation is given as,
Here,
The values of
Similarly, for
The value of
Similarly, for
With the help of the given data for oxygen-binding for lamprey hemoglobin, the plot between
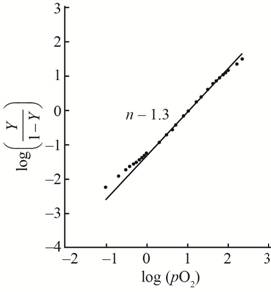
The value of slope
The hill coefficient for the graph is
(c)
Interpretation:
A model that is used to explain the observed cooperativity in oxygen binding by lamprey hemoglobin is to be stated.
Concept introduction:
Proteins are the biomolecules which are composed of the long chain of amino acid residues. The protein which contains oxygen and is present in the red blood cells in the body is known as hemoglobin. It contains iron as well.
Answer to Problem 16P
A model that is used to explain the observed cooperativity in oxygen binding by lamprey hemoglobin suggests that binding of oxygen with any monomer is easier as compared to binding of first oxygen with deoxygenated dimer.
Explanation of Solution
It is given that lamprey hemoglobin produces oligomers in the deoxygenated state. These oligomers were primarily dimmers in the deoxygenated state. These dimers are having low affinity for oxygen as compared to monomers. The cooperativity in oxygen binding is shown only if the dissociation of dimmers into two monomers occurs after the binding of first oxygen atom with a dimer. In this model, the binding of oxygen with every monomer is very easy as compared to the binding between first oxygen and the deoxy dimer.
Want to see more full solutions like this?
Chapter 7 Solutions
BIOCHEMISTRY (LOOSELEAF)-W/ACCESS
- You happen to pass a bar. Distracted by your entrance into the bar, one of the humans inadvertently drank a beverage containing 80 ml methanol. Unfortunately, no medical personnel are on the station, and you do not have access to sickbay. Assume that methanol and ethanol have densities of 0.79 g/ml, and that the alcohols distribute themselves rapidly over the total volume of 73 liters of body fluids of your host. A quick check of the biochemistry textbook that you always carry with you when traversing the promenade reveals that the Km of alcohol dehydrogenase for ethanol is 1 mM and for methanol the Km is 10 mM, and reminds you that alcohol dehydrogenase converts ethanol into acetaldehyde (which can then be converted to acetyl-CoA). Alcohol dehydrogenase also converts methanol into formaldehyde, which is quite toxic to humans. Ingesting 80 ml of methanol will permanently harm the human unless you intervene.b. Someone has a single 600 ml bottle of Romulan ale (52% ethanol), which he is…arrow_forwardAnimals produce carbon dioxide. Where does this carbon dioxide come from in the human body? Give the name of the processes where carbon dioxide is produced and explain how it is produced.arrow_forwardAnimals breathe in oxygen. What is this atmospheric oxygen used for in the human body? (Give the name of the process where atmospheric oxygen is used and explain what happens to the atmospheric oxygen molecule.)arrow_forward
- Please help me with this question. PLEase give me the right answer and for number 2 WRITE AT LEAST A PARAGRAPGH and dont copy from internet.arrow_forwardsurfactant. She has signs of low oxygen and high carbon dioxide. Explain why the lack of surfactant would result in respiratory distress. Furthermore, what type of acid-base disorder might she develop and how would her body compensate. Be detailed in your explanation and support your answer with facts from your textbook, research, and articles from scholarly journals. In addition, remember to add references in APA format to your posts to avoid plagiarism.arrow_forwardRespiratory paralysis. Tabun and sarin have been used as chemical-warfare agents, and parathion has been employed as an insecticide. What is the molec ular basis of their lethal actions? Tabun H3C Sarin -NO2 Parathionarrow_forward
- No free lunch. Explain why maintaining a high concentration of CO2 CO2 in the bundle-sheath cells of C4C4 plants is an example of active transport. How much ATP is required per CO2CO2 to maintain a high concentration of CO2CO2 in the bundle-sheath cells of C4C4 plants?arrow_forwardSituational task. The patient has a significant decrease in blood hemoglobin. No blood loss was observed. Diet and normalization of nutrition did not affect the increase in hemoglobin. What could be the cause of this phenomenon?arrow_forwardBe able to describe the hemoglobin binding curve and why it is S shaped. Why is the shape important for loading and off-loading Oxygen? What is subunit cooperativity?arrow_forward
- . Oxygen binding by the hemocyanin of the shrimp Callianassa has been measured. Using the following data, prepare a Hill plot and determine (a) Pso, (b) h (the Hill coefficient), and (c) the minimum number of oxygen binding sites on the protein molecule. Poz (mm Hg) Yoz Po, (mm Hg) Yo2 1.1 0.003 136.7 0.557 7.7 0.019 166.8 0.673 10.7 0.035 203.2 0.734 31.7 0.084 262.2 0.794 71.9 0.190 327.0 0.834 100.5 0.329 452.0 0.875 123.3 0.487 736.7 0.913arrow_forwardThe Cori Cycle. Before vigorous exercise (at rest) the level of blood lactate is at its normal level (about 25 mM). During a 400 m sprint, the value rises sharply in less than a few minutes to about 200 mM and then declines slowly to around 40 mM over 60 mins after the sprint. a. Discuss (or illustrate) the pathway and reactions that cause the rapid increase in lactate concentration during the sprint. b. What causes the slow decline in lactate concentration after the sprint? Why does the decrease occur more slowly than the rapid increase? c. What enzymatic reaction is responsible for maintaining the lactate concentration above zero at recovery after the sprint?arrow_forwardQuestion- Some of the reactions of NO in the blood do not cause problems at low concentrations but can upset the normal reactions of hemoglobin if there is a large concentration of NO. The same reactions can cause trouble if a synthetic blood substitute contains a molecule similar to hemoglobin but does not contain all the other enzymes normally contained in red blood cells. Explain what this problem is and how it arises.arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





