
Concept explainers
(a)
Interpretation:
From the given compounds, the compound with high regioselective ability towards addition of
Concept introduction:
Addition Reaction: It is defined as
The product of electrophilic addition reaction obtained by addition of electrophile to
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
Leaving group: it is a fragment that leaves substrate with a pair of electrons via heterolytic bond cleavage.
Chemical reaction involves bond making and breaking of two or more reactants in order to attain products from the reactants.
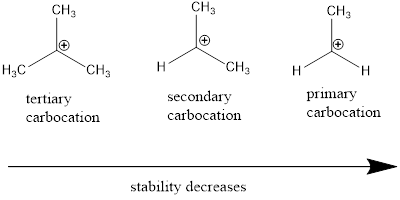
Cation: The positively charged chemical species is referred as cation.
Regioselective reaction: They are reactions which contain more than one product which are actually molecules with same molecular formula but different in the way they are connected and among those products only one product is major.
(b)
Interpretation:
From the given compounds, the compound with high regioselective ability towards addition of
Concept introduction:
Addition Reaction: It is defined as chemical reaction in which two given molecules combines and forms product. The types of addition reactions are electrophilic addition, nucleophilic addition, free radical additions and cycloadditions. Generally, compounds with carbon-hetero atom bonds favors addition reaction.
The product of electrophilic addition reaction obtained by addition of electrophile to
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
Leaving group: it is a fragment that leaves substrate with a pair of electrons via heterolytic bond cleavage.
Chemical reaction involves bond making and breaking of two or more reactants in order to attain products from the reactants.
Cation: The positively charged chemical species is referred as cation.
Regioselective reaction: They are reactions which contain more than one product which are actually molecules with same molecular formula but different in the way they are connected and among those products only one product is major.
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Essential Organic Chemistry (3rd Edition)
- Which compound has a larger Rf-value? Explainarrow_forwardWill the bonding of the Zn2+ ion to the ketone oxygen lone pair make theketone C=O easier or more difficult to reduce? Why?arrow_forwardWhat is the configuration of the chiral center in the bromoepoxide? How do you account for the stereoselectivity of this seven-step conversion?arrow_forward
- Draw all the resonance forms for the sigma complexes corresponding to bromination of aniline at the ortho, meta, and para positions.arrow_forwardBriefly explain why there are 5 non-carbonyl carbon peaks in the 13C NMR spectrum of the molecule in this molecule (picture attached) but there are 6 peaks in its 1H NMR spectrum. (hint - has to do with chirality)arrow_forwardChoose the structure that corresponds to the IR spectrumarrow_forward
- Draw the 1H NMR spectrum of Ethyl bromide or 1-Bromoethane. Show the splittingindicating which is singlet, which is doublet, etcarrow_forwardRhodamine B is useful dye prepared in a manner very similar to fluorescein. Due to the nitrogen donating groups, rhodamine B absorbs 550 nm light and fluoresces 580 nm light. a) What color would you expect for a rhodamine B solution? b) What is the color of the fluoresced (emitted light)?arrow_forward_______________ cleavage occurs when the halogen leaves as a halide during the first step of E1. homolytic cleavage heterolytic cleavagearrow_forward
- Why is the delocalization energy of pyrrole (21 kcal/mol) greater than that of furan (16 kcal/mol)?arrow_forwardWhy permanganate has 5 peaks in its UV-Visible spectrum between 600-500 nm?arrow_forwardPlease help! Rank the first two groups in order of SN1 sensitivity and the second group in order of SN2 sensitivity. Halide ranking advice appreciatedarrow_forward
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole


