
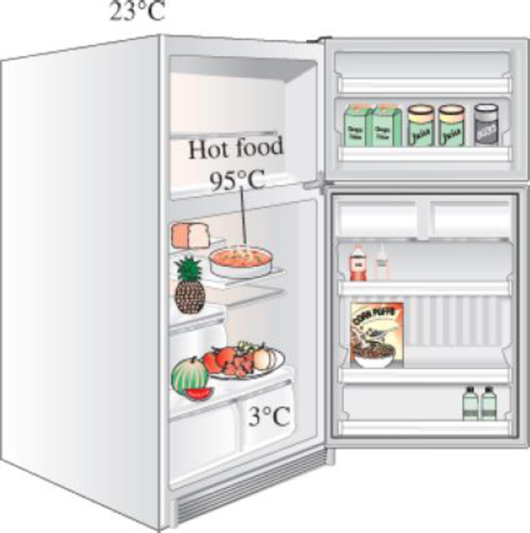
It is commonly recommended that hot foods be cooled first to room temperature by simply waiting a while before they are put into the refrigerator to save energy. Despite this commonsense recommendation, a person keeps cooking a large pan of stew three times a week and putting the pan into the refrigerator while it is still hot, thinking that the money saved is probably too little. But he says he can be convinced if you can show that the money saved is significant. The average mass of the pan and its contents is 5 kg. The average temperature of the kitchen is 23°C, and the average temperature of the food is 95°C when it is taken off the stove. The refrigerated space is maintained at 3°C, and the average specific heat of the food and the pan can be taken to be 3.9 kJ/kg·°C. If the refrigerator has a coefficient of performance of 1.5 and the cost of electricity is $0.125/kWh, determine how much this person will save a year by waiting for the food to cool to room temperature before putting it into the refrigerator.
FIGURE P6–123
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Thermodynamics: An Engineering Approach
- a) A gas is trapped in a piston-cylinder device. The initial pressure and temperature of the gas are 500 kPa and 300 K, respectively. The system undergoes an isothermal expansion process, in which 15KJ of boundary work is done by the gas and 3KJ of paddle-wheel work is done on the gas. Determine the amount heat transfer during this process. Explain the direction of the heat transfer. b) Air with a volume of 0.031 m³ is trapped in a piston-cylinder assembly at a temperature of 40°C and a pressure 102 kPa. The air is compressed adiabatically to a pressure 350 kPa. cv = 0.718 kJ/kg.k and cp = 1.00 kJ/kg. K Determine The work done during this process. c) As shown in Figure A1-c, a gas is contained in a piston-cylinder assembly. The mass and the cross-sectional area of piston are 3.20 kg and 35 cm², respectively. During heating, the spring is compressed and applies a force of 0.15 kN on the piston when the initial volume increased by 3 litres. Determine the final pressure in the cylinder…arrow_forwardA canister of sevoflurane, an inhaled anesthetic, is warmed to room temperature over the course of 30 minutes. As the gas warms, it pushes on a movable piston with a force of 623 mN, increasing the volume of the gas by 16 cm^3. How much work was done by the gas? A. 0.03 CJ B. 0.27 CJ C. 2.7 CJ D. 270 CJarrow_forward1. Is a temperature difference necessary to operate a heat engine? State why or why not. 2. Definitions of efficiency vary depending on how energy is being converted. Compare the definitions of efficiency for the human body and heat engines. How does the definition of efficiency in each relate to the type of energy being converted into doing work? 3. Why-other than the fact that the second law of thermodynamics says reversible engines are the most efficient-should heat engines employing reversible processes be more efficient than those employing irreversible processes? Consider that dissipative mechanisms are one cause of irreversibility. 1. (a) What is the efficiency of a cyclical heat engine in which 75.0 kJ of heat transfer occurs to the environment for every 95.0 kJ of heat transfer into the engine? (b) How much work does it produce for 100 k) of heat transfer into the engine? 2. The engine of a large ship does 2.00×10°J of work with an efficiency of 5.00%. (a) How much heat…arrow_forward
- (i) A can of soft drink at room temperature is put into the refrigerator so that it will cool. Would you model the can of soft drink as a closed system or as an open system? (ii) A candle is burning in a well-insulated room. Taking the room (the air plus the candle) as the system, determine if there is any heat transfer during the burning process. Explain how?arrow_forwardA used car salesman is trying to sell you a car by telling you that the cars engine, operating between 25 degrees Celsius and 125 degrees Celsius, has an efficiency of 82%. What is the efficiency of the car?arrow_forwardIf H is the work output of a heat engine and Q is the total heat input of an engine, then determine its thermal efficiency.arrow_forward
- The label on a washing machine indicates that the washer will use $85 worth of hot water if the water is heated by a 90 percent efficient electric heater at an electricity rate of $0.125/kWh. If the water is heated from 18 to 45°C, the amount of hot water an average family uses per year is (a) 19.5 tons (b) 21.7 tons (c) 24.1 tons (d) 27.2 tons (e) 30.4 tonsarrow_forwardDescribe the heat and work are energy transfer mechanisms between a system and its surroundings, and there are many similarities between them?arrow_forwardA house is to be maintained at 20°C when the outside air is at -25°C. It is determined that 1800 kJ is required each minute to accomplish this.If an electric heat pump is used to supply this energy, determine the minimum theoretical work input for one day of operation, in kJ/day. Evaluating electricity at 13 cents per kWh, determine the minimum theoretical cost to operate the heat pump, in $/day. ($1 = 100 cents)If a heat pump whose coefficient of performance is 3.0 provides the required heating, determine the operating cost, in $/dayarrow_forward
- A heat engine must provide 1000 Watts of power. Assuming it has an efficiency of 0.35, how much heat must be rejected by the engine, in Watts?arrow_forwardA piston cylinder assembly contains steam. Initially, the specific internal energy (internal energy per unit mass) is equal to 2600 kJ/kg. The steam undergoes a process during which 90 kJ of heat is transferred to the steam. On the other hand, using a specific mechanism installed in the cylinder, 20 kJ of energy are transferred to the steam by work. If 10 kJ of heat is dissipated from the steam to the outside of the cylinder through the wall of the cylinder, Determine the work done by the steam on the piston. The specific internal energy at the final state is equal to 2500 kJ/kg. The mass of the steam is 4.4 kg. Select one: a. 1211 kJ b. 540 kJ c. 780 kJ d. 328 kJarrow_forwardBy supplying energy to a house at a rate of 25,000 kJ/hr, a heat pump maintains the temperature of the dwelling at 20 C when the outside air is at -10 C. If electricity costs 8 cents per kW-hr, determine the minimum theoretical operating cost to heat the house for 24 hours. $1.97 O $1.37 $1.75 O $1.51 O$1.64arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





