
Concept explainers
Interpretation:
Pressure in each container before and after the stopcock is opened has to be calculated. Also, the container after the stopwatch is opened has to be drawn.
Concept Introduction:
The properties that are dependent upon the size or the amount of eth matter contained in the system are termed as extensive properties. They are additive in nature.
Any gas obeys the assumption laid down in kinetic molecular theory is said to be an ideal gas. The combination of all the
Here,
Answer to Problem 6.92QE
Pressure in the left and right container before the stopcock is opened are
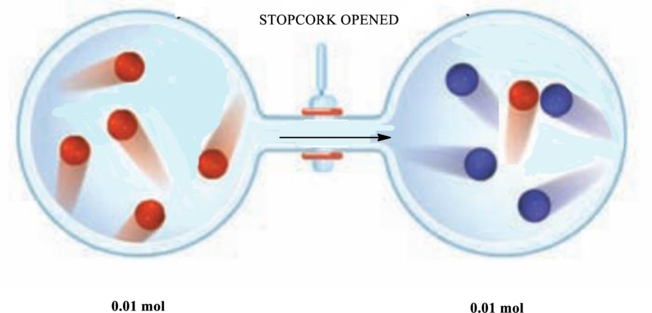
Explanation of Solution
Since each dot represents
Similarly, since there are four blue molecules that represent
The formula to convert degree Celsius to kelvin is as follows:
Here,
Substitute
Rearrange equation (1) to obtain the expression of temperature as follows:
Substitute
Substitute
After the stopcock is opened the volume is taken as the sum of the volume of both containers as it is an extensive property. Thus the total volume is calculated as follows:
Here,
Substitute
Similarly, the amount of substance or the number of moles is also an additive property, therefore, the total number of moles
Here,
Substitute
Substitute
Since when the stop corks are opened the number of moles must be equilibrated as the volume is equal on both sides. As a result, one molecule from the left container moves to the right so that each side has
Want to see more full solutions like this?
Chapter 6 Solutions
Chemistry: Principles and Practice
- A collapsed balloon is filled with He to a volume of 12.5 L at a pressure of 1.00 atm. Oxygen, O2, is then added so that the final volume of the balloon is 26 L with a total pressure of 1.00 atm. The temperature, which remains constant throughout, is 21.5 C. (a) What mass of He does the balloon contain? (b) What is the final partial pressure of He in the balloon? (c) What is the partial pressure of O2 in the balloon? (d) What is the mole fraction of each gas?arrow_forwardA mixture at 33 °C contains H2at 325 torr. N;at 475 tore and O2at 650. torr. What is the total pressure of the gases in the system? Which gas contains the greatest number of moles?arrow_forwardA 1 007-g sample of an unknown gas exerts a pressure of 715 mm Hg in a 452-mL container at 23 C. What is the molai mass of the gas?arrow_forward
- If equal masses of O2 and N2 are placed in separate containers of equal volume at the same temperature, which of the following statements is true? If false, explain why it is false. (a) The pressure in the flask containing N2 is greater than that in the flask containing O2. (b) There are more molecules in the flask containing O2 than in the flask containing N2.arrow_forwardConsider the following sketch. Each square in bulb A represents a mole of atoms X. Each circle in bulb B represents a mole of atoms Y. The bulbs have the same volume, and the temperature is kept constant. When the valve is opened, atoms of X react with atoms of Y according to the following equation: 2X(g)+Y(g)X2Y(g)The gaseous product is represented as and each represents one mole of product. (a) IfP A=2.0 atm, what is P8 before the valve is opened and the reaction is allowed to occur? What is P A+P B? (b) Redraw the sketch to represent what happens after the valve is opened. (c) What is PA? What is PB? What is P A+P B? Compare your answer with the answer in part (a).arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning





