
Concept explainers
Interpretation:
By using Lewis’s theory the covalent bond has to be explained.
Concept introduction:
Lewis theory is about
- Ionic bonding(complete transfer of electrons)
- Covalent bonding (sharing of electrons)
Answer to Problem 6.1QP
The Covalent bond is formed by the sharing of electron pair between two atoms by using Lewis’s theory.
Explanation of Solution
- Covalent Bonding
In covalent bonding the atoms share electrons equally, it means both the atoms have an equal share of pair of electrons in the bond, and this sharing of electrons creates a bond that is said to be covalent bond.
- Explain sharing of electron pair between two atoms:
Let us discuss an example for the formation of covalent bond in fluorine.
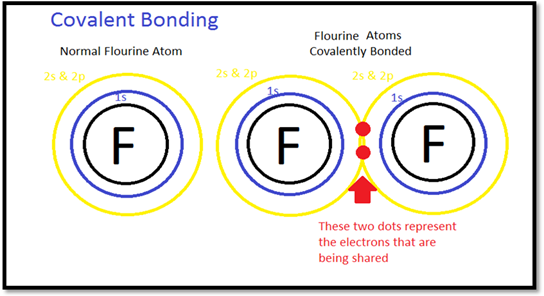
According to Lewis’s theory, covalent bond is formed by the sharing of electron pair between two atoms. As shown in figure, fluorine has two atoms each fluorine atom shares electrons to get electron pair which is represented in two dots. This electron pair forms the bond is known as covalent bond.
By using Lewis’s theory the covalent bond was explained.
Want to see more full solutions like this?
Chapter 6 Solutions
Chemistry Atoms First, Second Edition
- hy is there an octet rule (and what does actet mean) in writing Lewis structures?arrow_forwardWrite the Lewis structure for nitrosyl fluoride, FNO. Using only a periodic table, identify (a) which is the longer bond. (b) which is the stronger bond. (c) which is the more polar bond.arrow_forwardWhat is the difference between a Lewis dot symbol and a Lewis structure?arrow_forward
- Explain how the concept of formal charge can be used to identify the dominant Lewis structure?arrow_forwardThe chemical formula of methane is CH4. The Lewis Structure of methane is shown below. Explain how a chemical formula differs from a Lewis structure. H-C-Harrow_forwardThe table below includes the bond enthalpy (the energy required to separate the diatomic molecule into its atoms) and the bond length for each diatomic molecule. Diatomic Molecule Bond Enthalpy (kJ/mol) Bond Length (pm) Cl2 243 199 O2 498 121 N2 945 110 Identify the observed trend between bond enthalpy and number of shared electrons: as the number of electrons shared between two atoms increases, the bond enthalpy ( increases / decreases / remains unchanged ). Identify the observed trend between bond length and number of shared electrons: as the number of electrons shared between two atoms increases, the bond length ( increases / decreases / remains unchanged ).arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

 Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER  Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning





