
Concept explainers
Interpretation: The structure for the exo product formed by the reaction of cyclopentadiene with maleic anhydride should be predicted.
Concept introduction:
The reaction between a diene (4π electron system) and a dienophile (2π electron system) to give a substituted cyclohexane derivative is termed as Diels Alder reaction. This reaction is a cycloaddition reaction.
Answer to Problem 1Q
The structure for the exo product formed by the reaction of cyclopentadiene with maleic anhydride is represented as follows:
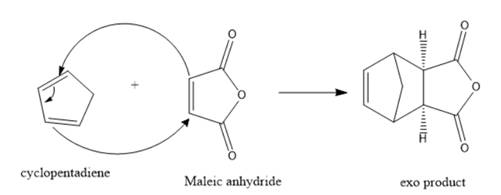
Explanation of Solution
Cyclopentadiene is a 4π electron system thus, it will be the diene. On the other hand, maleic anhydride is a 2π electron system thus, it will act as a dienophile. Therefore, the product formation for the reaction between cyclopentadiene and maleic anhydride should be represented as follows:
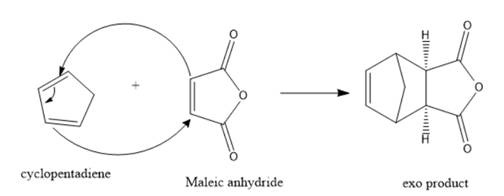
Want to see more full solutions like this?
Chapter 52 Solutions
A Small Scale Approach to Organic Laboratory Techniques
- Write the product of the following reaction. Indicate the mechanism of the reaction involved.arrow_forwardA compound of molecular formula C5H10O forms a yellow precipitate with 2,4-dinitrophenylhydrazine reagent and a yellow precipitate with reagents for the iodoform test. a) Draw the structural formulae b) Name the two (2) compounds that fit these tests.arrow_forwardDraw the structure of the major product of 2-methyl-3-heptyne + H2, Lindlar catalystarrow_forward
- What three alkenes (excluding stereoisomers) can be used to prepare 3chloro-3-methylhexane by addition of HCl?arrow_forwardWhat is the major organic product obtained from the reaction of toluene with fuming sulfuric acid?arrow_forwardSpecialized reagents and their acronyms: MCPBA= meta-chloroperbenzoic acid; HOAC = acetic acid, NaOAc = Sodium Acetate; Ph= phenyl = C6H5-; P(Ph)3 = triphenyl phosphine; NBS = N- bromosuccinimide, PBR3, SOCI2, AlCl3, FeBr3, H2SO4, Li, Na, Mg, Br2, CrO3, LDA = lithium diisopropylamide, PCC = pyridinium chlorochromate, KO-C(CH3)3, LAH = LIAIH4, (sia)2BH = disecondary isobutyl borane, KMNO4, HIO4 = periodic acid %3D %3D %3D %3D %3D write the missing reagents and solvents over the arrows. Some transformation require multiple steps. In some cases there will be multiple arrows. Br Brarrow_forward
- Specialized reagents and their acronyms: MCPBA= meta-chloroperbenzoic acid; HOAC = acetic acid, NaOAc = Sodium Acetate; Ph= phenyl = C6H5-; P(Ph)3 = triphenyl phosphine; NBS = N- bromosuccinimide, PBR3, SOCI2, AICI3, FeBr3, H2SO4, Li, Na, Mg, Br2, CrO3, LDA = lithium diisopropylamide, PCC = pyridinium chlorochromate, KO-C(CH3)3, LAH = LIAIH4, (sia)2BH = disecondary isobutyl borane, KMNO4, HIO4 = periodic acid %3D write the missing reagents and solvents over the arrows. Some transformation require multiple steps. In some cases there will be multiple arrows. Ph -Ph Ph Ph Brarrow_forwardSpecialized reagents and their acronyms: MCPBA= meta-chloroperbenzoic acid; HOAC = acetic acid, NaOAc = Sodium Acetate; Ph= phenyl = C6H5-; P(Ph)3 = triphenyl phosphine; NBS = N- bromosuccinimide, PBR3, SOCI2, AICI3, FeBr3, H2SO4, Li, Na, Mg, Br2, CrO3, LDA = lithium diisopropylamide, PCC = pyridinium chlorochromate, KO-C(CH3)3, LAH = LIAIH4, (sia)2BH = disecondary isobutyl borane, KMNO4, HIO4 = periodic acid %3D %3D %3D %3D %3D %3D %3D write the missing reagents and solvents over the arrows. Some transformation require multiple steps. In some cases there will be multiple arrows. ОН H. (R) Br (S) HO OHarrow_forwardSpecialized reagents and their acronyms: MCPBA= meta-chloroperbenzoic acid; HOAC = acetic acid, NaOAc = Sodium Acetate; Ph= phenyl = C6H5-; P(Ph)3 = triphenyl phosphine; NBS = N- bromosuccinimide, PBr3, SOCI2, AICI3, FeBr3, H2SO4, Li, Na, Mg, Br2, CrO3, LDA = lithium diisopropylamide, PCC = pyridinium chlorochromate, KO-C(CH3)3, LAH = LIAIH4, (sia)2BH = disecondary isobutyl borane, KMNO4, HIO4 = periodic acid %3D %3D %3D %3D %3D write the missing reagents and solvents over the arrows. Some transformation require multiple steps. In some cases there will be multiple arrows. Ph CH CH o 0om ilubongo CEC-Ph In questions 14-24, write the products of the following transformations. If there is no reaction write NR. Indicate R or S or racemic or indicate cis or trans where appropriate OH + LDA THF solvent (S) Pharrow_forward
- Restate the paragraph. The main objective of the laboratory activity was to make aspirin also known as acetylsalicylic acid in its scientific name. It was successfully done as aspirin was synthesized using the combination of salicylic acid and acetic anhydride. In this reaction, salicylic acid's hydroxyl group on the benzene ring interacted with acetic anhydride to generate an ester functional group. This is an esterification reaction that produces acetylsalicylic acid. Sulfuric acid was utilized as a catalyst to start the esterification reaction. Some unreacted acetic anhydride and salicylic acid, as well as sulfuric acid, aspirin, and acetic acid, remained in the solution after the reaction was completed. The process of crystallization was used to obtain pure crystals of a substance from an impure mixture. Upon completion of the lab, analysis, and calculations, , it is evident that the synthesis of aspirin is possible using these methods but that the yield will be relatively lowarrow_forwardWhy does the addition of bromine to fumaric acid require a high temperature for the reaction while other substrates (generic alkanes, alkenes, and alkynes) react with bromine at room temperature?arrow_forwardDraw the structure of the prepolymer A formed from 1,4-dihydroxybenzene and excess epichlorohydrin.arrow_forward
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
