
Concept explainers
Arrange the following pairs of charged particles in order of increasing electrostatic potential energy.
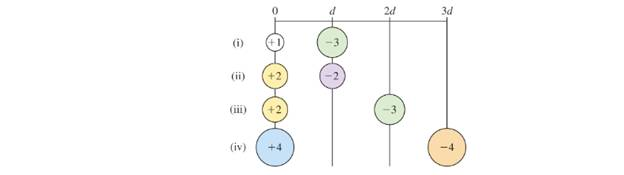
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Chemistry
Additional Science Textbook Solutions
EBK INTRODUCTION TO CHEMISTRY
Chemistry
Introduction to Chemistry
Chemistry: The Central Science (13th Edition)
Organic Chemistry - Standalone book
Thermodynamics, Statistical Thermodynamics, & Kinetics
- 18 - for Cl, Cl + and Cl- ions;Cl- is the largest number of electronsII. Stripping electrons from the Cl + ion is most difficultIII. Neutral Cl atom and Cl- ions have equal radiiWhich of his judgments are correct? A) Only IIIB) I, IIC) I, IIID) I onlyE) I, II, IIIarrow_forwardBetween Na and Ne, which one has higher first ionization energy and why? Make sure to discuss effective nuclear charge, forces, and energy. (2-3 sentences)arrow_forwardWhat is a resonance dot structure? How do you know if you have resonance? I !!! H Normal EEAA Enter your answer here ||' ||| ||| BIUS √x 2 T₂arrow_forward
- In(s) + Fe(s) -> FeI,(s) Match the words in the left column to the appropriate blanks in the sentences on the right.arrow_forwardM Inbox (6)- danasia.... 31 Ascension - Calend... ||| O Advanced Material Predicting the relative lattice energy of binary ionic compounds In each row, pick the compound with the bigger lattice energy. Note: lattice energy is always greater than zero. Which compound has the bigger lattice energy? CaBr₂ Cal₂ O O Cs Cl FH Explanation tab X Esc O Rb₂0 Check Type here to search ! 1 F1 Q F2 Shared drives - Goo... Intrane @ 2 W t F3 # m 3 H F4 $ E 4 & F5 % 5 Rarrow_forward19 of 44 > © Macmillan Learning In the given three-dimensional molecular structure, the differently colored spheres represent different types of atoms. Write a molecular formula for this molecule. molecular formula: 4 R % 67 68 5 1 SPECIAL xº (g) ΔΣΩ λμπ X₁ X () 6 [] (s) Y (1) (aq) →>>> MacBook Pro The 11 7 U t CLR 8 K →>> 9 Rotate X You 9 Rotate Y ( 0 ☐C □H D Rotate Z 0 Zoom In P O Zoom Out Attempt A Label Atoms Carrow_forward
- Part B The dipole moment (u) of HBr (a polar covalent molecule) is 0.844D (debye), and its percent ionic character is 12.5 % . Estimate the bond length of the H Br bond in picometers. Note that • 1D= 3.34 x 10-30 C .m and • in a bond with 100% ionic character, Q = 1.6 x 10-19 C. Express your answer to two significant figures and include the appropriate units. HA ? r= 5.3 • 1018 pmarrow_forwardN Calculate the lattice enthalpy for RbCl. You will need the following information: Species A,H, kJ/mol Rb(e) RbCl() CI(g) Enthalpy of ionization for Rb(g) is 403.0 kl/mol; electron attachment enthalpy for Cl(g) is-349.0 kJ/mol Lattice enthalpy kJ/mol Submit Answer *** 1 43 E D 80.9 -435.4 121.3 Try Another Version 80 54 R F di a 25 2 item attempts remaining T G ‹6 P FA Y & 29 7 H 8 #7 U 8 Dil FA - 6 8 Karrow_forwardConstruct a Born–Haber cycle for the formation of the hypotheticalcompound NaCl2, where the sodium ion has a 2+charge (the second ionization energy for sodium is given inTable 7.2). (a) How large would the lattice energy need to befor the formation of NaCl2 to be exothermic? (b) If we wereto estimate the lattice energy of NaCl2 to be roughly equalto that of MgCl2 (2326 kJ/mol from Table 8.1), what valuewould you obtain for the standard enthalpy of formation,ΔHf°, of NaCl2?arrow_forward
- (A) if both sentences are true, (B) if both sentences are false, (C) if the first sentence is true but the second is false, and (D) if the first sentence is false but the second is true. 1. Water appears in nature in all three common states of matter. Water is a tasteless, odorless liquid at standard temperature and pressure. 2. The addition of salt in the distilling mixture made the water impure and saline. The addition of 5 drops of the silver nitrate solution into the distillate determines whether the distillate is impure or pure water. 3. The ideal electrical conductivity of totally pure water is 0.055 microSiemens, whereas typical deionized water has an electrical conductivity of 0.1 microSiemens. The freezing point of water at 1 atm is 100degC. 4. From the air, oxygen can slowly diffuse across the water’s surface from the surrounding atmosphere, or be mixed in quickly through aeration, whether natural or man-made. The aeration of water can be caused by wind (creating waves),…arrow_forward(ii) Construct and label a Born-Haber cycle for strontium difluoride, SrF2. Calculate the lattice energy of SrF2 using the following data: First electron affinity of fluoride =–328.0 kJ mol"1 First ionization energy of strontium = 549.0 kJ mol" Second ionization energy of strontium = 1064.0 kJ mol"! Enthalpy of atomization of fluoride = 75.3 kJ mol" Enthalpy of atomization of strontium = 164.0 kJ mol"! Enthalpy of formation of strontium difluoride = -1224.4 kJ mol"!arrow_forwardALEKS - Iffa X FSA Documents X M Direct PLUS X FSA Aid Summa X FSA www-awn.aleks.com/alekscgi/x/lsl.exe/1o_u-IgNslkr7j8P3jH-lijkPWvZoZLqKt1FLlq7wcPWKzBYGFE9IMFjNv26Nxc O ATOMS, IONS AND MOLECULES Naming ionic compounds with common polyatomic ions Fill in the name and empirical formula of each ionic compound that could be formed from the ions in this table: Some ionic compounds cation anion empirical formula name of compound Cro 2+ Zn 2+ Pb CN 2+ Fe C,H,O,arrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning

