
Organic Chemistry
5th Edition
ISBN: 9780078021558
Author: Janice Gorzynski Smith Dr.
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5, Problem 5.7P
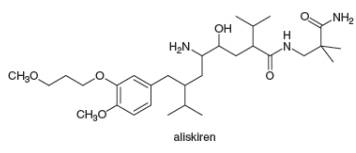
The facts in section 5.4A can be used to locate stereogenic centers in any molecule, no matter how complicated. Always look for carbons surrounded by four different groups. With this in mind, locate the four stereogenic centers in aliskiren, a drug introduced in
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
1a. How many stereogenic centers are present 1c. Draw a three-dimensional structure of a
in the structure below? Indicate them with
asterisk(s). How many stereoisomers
stereoisomers are
possible?
chiral compound with the molecular formula
of C4H4Cl₂ that does not have a stereogenic
carbon. In addition, draw the enantiomer of
this compound.
Number of stereogenic centers:
Number of stereoisomers possible:
1b. Draw one of the two most stable
stereoisomers of the compound in 1a using a
planar structure with wedges and dashes. Now
draw it in its preferred chair conformation.
1d. Draw two meso compounds with the
molecular formula of C7H14.
2. The molecules shown below are four terpineol isomers. They are
isolated from plants and have distinct aromas. For example, a -terpineol
is a common perfume ingredient and smells similar to lilacs.
Hol
H-O-
a-terpineol
B-terpineol
Y-terpineol
4-terpineol
A) Draw a structural isomer of terpineol that meet the following criteria. You should draw one unique structure for each set. You
must draw each molecule twice, once as a skeletal structure and once as a complete Lewis structure showing all atoms, bonds,
and lone pairs of electrons.
i. Does not have a ring.
ii. Has an E alkene.
iii. Has a secondary alcohol.
iv. Does not contain an alcohol.
B) Draw a complete Lewis structure of a-terpineol showing all bonds, atoms and lone pairs of electrons.
Q1: The shrub ma huang contains two biologically active stereoisomers ephedrine
and pseudoephedrine-with two stereogenic centers as shown in the given
structure. Ephedrine is one component of a once popular combination drug used
by body builders to increase energy and alertness, while pseudoephedrine is a
nasal decongestant.
a. Draw the structure of naturally occurring (-)-ephedrine,
which has the 1R, 25 configuration.
b. Draw the structure of naturally occurring (+)-
pseudoephedrine, which has the 15,25 configuration.
c. How are ephedrine and pseudoephedrine related? N,
d. Draw all other stereoisomers of (-)-ephedrine and
(+)-pseudoephedrine and give the R,S designation for all
stereogenic centers.
OH
Ephedrine
15 2.5
Chapter 5 Solutions
Organic Chemistry
Ch. 5 - Prob. 5.1PCh. 5 - Prob. 5.2PCh. 5 - Draw the mirror image of each compound. Label each...Ch. 5 - Prob. 5.4PCh. 5 - A molecule is achiral if it has a plane of...Ch. 5 - Problem 5.6 Locate the stereogenic centers in each...Ch. 5 - The facts in section 5.4A can be used to locate...Ch. 5 - Prob. 5.8PCh. 5 - Prob. 5.9PCh. 5 - Prob. 5.10P
Ch. 5 - Prob. 5.11PCh. 5 - Prob. 5.12PCh. 5 - Label each compound as R or S.Ch. 5 - Prob. 5.14PCh. 5 - Prob. 5.15PCh. 5 - Prob. 5.16PCh. 5 - Prob. 5.17PCh. 5 - Problem 5.18 Compounds E and F are two isomers of...Ch. 5 - Prob. 5.19PCh. 5 - Prob. 5.20PCh. 5 - Prob. 5.21PCh. 5 - Prob. 5.22PCh. 5 - Prob. 5.23PCh. 5 - Prob. 5.24PCh. 5 - Which of the following cyclic molecules are meso...Ch. 5 - Prob. 5.26PCh. 5 - Prob. 5.27PCh. 5 - Problem 5.28 The amino acid has the physical...Ch. 5 - Prob. 5.29PCh. 5 - Prob. 5.30PCh. 5 - Prob. 5.31PCh. 5 - Prob. 5.32PCh. 5 - Prob. 5.33PCh. 5 - Prob. 5.34PCh. 5 - Prob. 5.35PCh. 5 - Prob. 5.36PCh. 5 - Prob. 5.37PCh. 5 - Prob. 5.38PCh. 5 - Prob. 5.39PCh. 5 - 5.40 Determine if each compound is identical to or...Ch. 5 - Prob. 5.41PCh. 5 - Prob. 5.42PCh. 5 - Prob. 5.43PCh. 5 - Prob. 5.44PCh. 5 - Prob. 5.45PCh. 5 - Prob. 5.46PCh. 5 - Label each stereogenic center as R or S. a. c. e....Ch. 5 - Prob. 5.48PCh. 5 - Prob. 5.49PCh. 5 - Prob. 5.50PCh. 5 - Prob. 5.51PCh. 5 - Prob. 5.52PCh. 5 - Prob. 5.53PCh. 5 - Prob. 5.54PCh. 5 - Prob. 5.55PCh. 5 - Prob. 5.56PCh. 5 - Prob. 5.57PCh. 5 - Prob. 5.58PCh. 5 - 5.59 Explain each statement by referring to...Ch. 5 - Prob. 5.60PCh. 5 - Prob. 5.61PCh. 5 - Prob. 5.62PCh. 5 - Prob. 5.63PCh. 5 - Prob. 5.64PCh. 5 - Prob. 5.65PCh. 5 - Prob. 5.66PCh. 5 -
5.67 Artemisinin and mefloquine are widely used...Ch. 5 - 5.68 Saquinavir (trade name Invirase) is a...Ch. 5 - Prob. 5.69PCh. 5 - Prob. 5.70PCh. 5 - Prob. 5.71PCh. 5 - Prob. 5.72PCh. 5 - Problem 5.73 An acid-base reaction of with a...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Q1: The shrub ma huang contains two biologically active stereoisomers ephedrine and pseudoephedrine-with two stereogenic centers as shown in the given structure. Ephedrine is one component of a once popular combination drug used by body builders to increase energy and alertness, while pseudoephedrine is a nasal decongestant. a. Draw the structure of naturally occurring (-)-ephedrine, which has the 1R, 25 configuration. b. Draw the structure of naturally occurring (+)- pseudoephedrine, which has the 15,25 configuration. C. How are ephedrine and pseudoephedrine related? , d. Draw all other stereoisomers of (-)-ephedrine and (+)-pseudoephedrine and give the R,S designation for all stereogenic centers. OH Ephedrine 825 1S 25arrow_forwardLocate the stereogenic centers in each molecule. Compounds may have one or more stereogenic centers.arrow_forwardWhat is the name for a 50:50 mixture of enantiomers such as a 50:50 mixture of (S)-2- butanol and (R)-2-butanol? Conformers, racemic, diastereomers, meso, different, stereopure, chiral, optically activearrow_forward
- Indicate whether the pair of structures shown represent stereoisomers, constitutional isomers, different conformations of the same compound, or the same conformation of a compound viewed from a different perspective. Note that cis, trans isomers are an example of stereoisomers. OH Но. Но. CH3 CH3 H3C- H3C- HO OHarrow_forwardDraw a three-dimensional structure for each compound, and star all asymmetric carbon atoms. Draw the mirror image for each structure, and state whether you have drawn a pair of enantiomers or just the same molecule twice. Build molecular models of any of these examples that seem difficult to you. 1-bromo-2-methylbutanearrow_forwardIndicate whether the pair of structures shown represent stereoisomers, constitutional isomers, different conformations of the same compound, or the same conformation of a compound viewed from a different perspective. Note that cis, trans isomers are an example of stereoisomers. H. H. Cl H. CI CH CH3 но.arrow_forward
- There are nine constitutional isomers of molecular formula C7H16. 1) What is the unsaturation number of each of these compounds? 2) Draw five constitutional isomers, where two of these are chiral compounds. 3) For the three achiral constitutional isomers, provide the correct IUPAC names. 4) For the two chiral constitutional isomers, draw in Fischer projection formula the enantiomers of each. 5) Name, according to IUPAC standards, each of the enantiomers you drew in #4 above.arrow_forward1. Draw all of the stereoisomers of 2,4-dichloropentane. Indicate the relationship between these stereoisomers, such as enantiomers, diastereomers, or meso compounds.arrow_forwardFill in the boxes with the correct organic structure. You must show relative stereochemistry where necessary. You only need to draw one enantiomer if the product is a racemic mixture. 1. OsO4 2. NaHSO3 MCPBA HglO6 1. NaOH 2. HCIarrow_forward
- Indicate whether the pair of structures shown represent stereoisomers, constitutional isomers, different conformations of the same compound, or the same conformation of a compound viewed from a different perspective. Note that cis, trans isomers are an example of stereoisomers. Но. CI Но- CH3 H3C- H3C- ОН OHarrow_forwardIndicate whether the pair of structures shown represent stereoisomers, constitutional isomers, different conformations of the same compound, or the same conformation of a compound viewed from a different perspective. Note that cis, trans isomers are an example of stereoisomers. CH3 CI- CH3 CH3- H3C- CH3- 오. CH3 ♦arrow_forwardLocate the tetrahedral stereogenic center(s) in each compound. A molecule may have one or more stereogenic centers.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License