The reaction occurring in the titration is the neutralization of OH− (from Ba(OH)2) by H3O+ (from HCl):
Ba(OH)2(aq) + 2HCl(aq) → 2H2O(l) + BaCl2(aq)H3O+(aq) + OH−(aq) → 2H2O(l)
Which shows that 2 equivalent of acid react with 1 equivalent of Barium oxide. For achieving equivalence point, we need the equivalent quantities of acid and base. So
M1V1 = M2V2
Where,
M1= Concentration of base
M2= Concentration of acid
V1= Volume of base
V2= Volume of acid
Substituting the given values in the above equation and solving for V2,
M1V1 = M2V2(0.15M)(25 mL)= (2×0.22)(V2)V2= (0.15M)(25 mL)2×0.22= 8.522 mL.
For neutralization we need 8.522 mL of acid.
For plotting the graph we can start the titration by taking 0.00mL of acid and gradually add acid in dropwise manner till 8.5 mL.
Case – 1:
Given data:
Volume of Ba(OH)2 = 25.00 ml = 25.00×10−3LConcentration of Ba(OH)2 = 0.15MConcentration of HCl = 0.22MVolume of HCl added = 0.00 ml = 0.00×10−3LThe total volume of the solution at this point = 25.00×10−3L + 0.00×10−3L = 25.00×10−3L
The initial number of moles of Ba(OH)2= Molarity×Volume = (0.15 M)(25.00×10−3L) = 3.75×10−3.
Moles of added HCl = Molarity×Volume = (0.22M)(0.00×10−3L) =0.
No. of moles Ba(OH)2(aq) + 2HCl(aq) → 2H2O(l) + BaCl2(aq)_ Initial 3.75×10−3 0 − 0Change 0 0 − 0_Final 3.75×10−3 0 − 0
Excess[OH−] = 3.75×10−3 moles25.00×10−3L = 0.15 M.
pOH=−log[OH−] = −log(0.15) = 0.8239.
pH = 14 − pOH = 14−0.8239=13.1761.
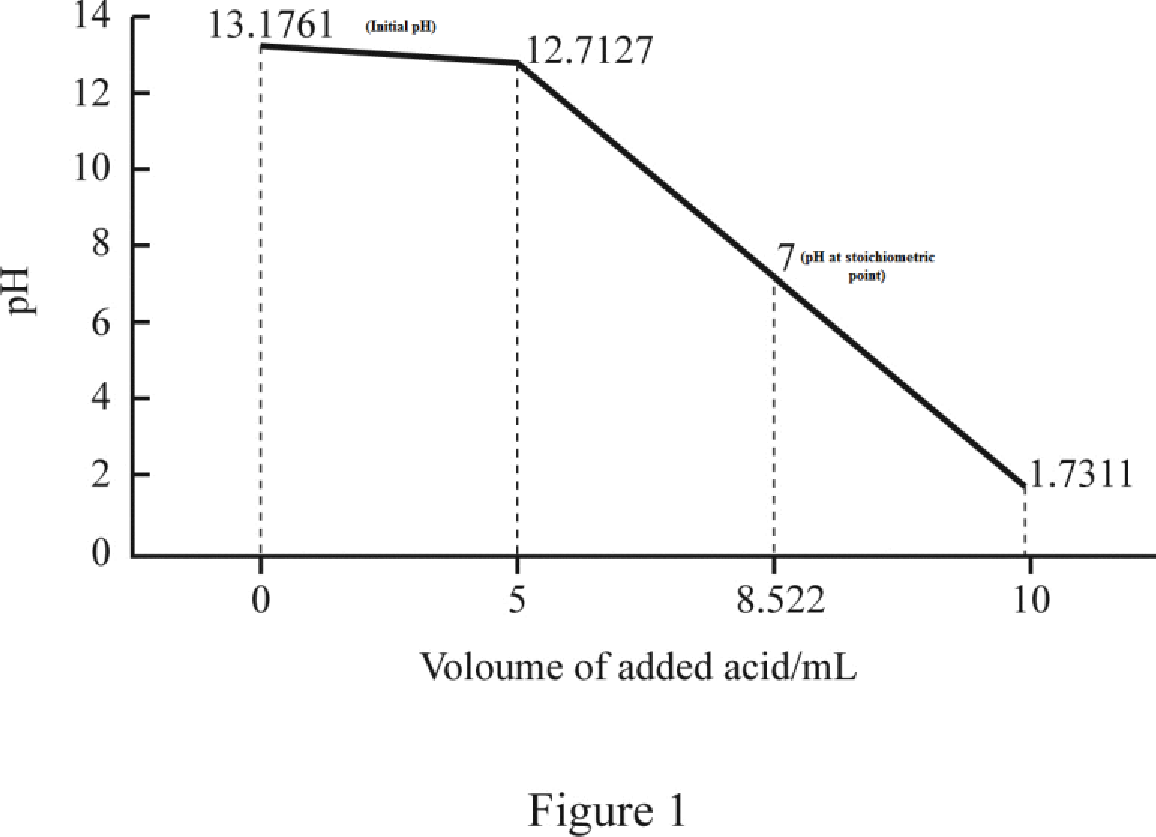
Therefore, the initial pH of the solution has been calculated to be 13.1761.
Case – 2:
Volume of HCl added = 5.00 ml = 5.00×10−3LThe total volume of the solution at this point = 25.00×10−3L + 5.00×10−3L = 30.00×10−3L
Moles of added HCl = Molarity×Volume = (0.22M)(5.00×10−3L) = 1.1×10−3.
As there is two molecules of HCl, moles of added HCl will be 2.2×10−3.
No. of moles Ba(OH)2(aq) + 2HCl(aq) → 2H2O(l) + BaCl2(aq)_ Initial 3.75×10−3 2.2×10−3 − 0Change −2.2×10−3 − 2.2×10−3 − +2.2×10−3_Final 1.55×10−3 0 − 2.2×10−3
Excess[OH−] = 1.55×10−3 moles30.00×10−3L = 0.0516M.
pOH=−log[OH−] = −log(0.0561) = 1.2873.
pH = 14 − pOH = 14−1.2873=12.7127.
Therefore, the pH of the solution has been calculated to be 12.7127.
Case – 3:
Volume of HCl added = 8.522 ml = 8.522×10−3LThe total volume of the solution at this point = 25.00×10−3L + 8.522×10−3L = 33.522×10−3L
Moles of added HCl = Molarity×Volume = (0.22M)(8.522×10−3L) = 1.8748×10−3.
As there is two molecules of HCl, moles of added HCl will be 3.75×10−3.
No. of moles Ba(OH)2(aq) + 2HCl(aq) → 2H2O(l) + BaCl2(aq)_ Initial 3.75×10−3 3.75×10−3 − 0Change −3.75×10−3 − 3.75×10−3 − +3.75×10−3_Final 0 0 − 3.75×10−3
The HCl will react with an equal amount of the Ba(OH)2 and 0.0 mol Ba(OH)2 will remain. This is the equivalence point of a weak base –strong acid titration, thus, the pH is 7.0. Only the neutral salt BaCl2 is in solution at the equivalence point.
Case – 4:
Volume of HCl added = 10.00 ml = 10.00×10−3LThe total volume of the solution at this point = 25.00×10−3L + 10.00×10−3L = 35.00×10−3L
Moles of added HCl = Molarity×Volume = (0.22M)(10.00×10−3L) = 2.2×10−3.
As there is two molecules of HCl, moles of added HCl will be 4.4×10−3.
No. of moles Ba(OH)2(aq) + 2HCl(aq) → 2H2O(l) + BaCl2(aq)_ Initial 3.75×10−3 4.4×10−3 − 0Change −3.75×10−3 − 3.75×10−3 − +3.75×10−3_Final 0 0.65×10−3 − 3.75×10−3
Excess[H3O+] = 0.65×10−3 moles35.00×10−3L = 0.0185 M.
pH=−log[H3O+] = −log(0.0185) = 1.7311.
Therefore, the pH of the solution has been calculated to be 1.7311.
Now we can plot the graph as shown in figure 1.
Volume of added acid/mLpH013.1761512.71278.5227.0000101.7311


 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY




