
CHEMISTRY-TEXT
8th Edition
ISBN: 9780134856230
Author: Robinson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5, Problem 5.27CP
The following diagram shows the energy levels of the different shells in the hydrogen atom.
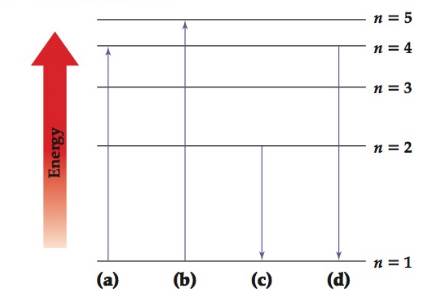
(a) Which transition corresponds to absorption of light with the longest wavelength?
(b) Which transition corresponds to emission of light with the shortest wavelength?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
CHEMISTRY-TEXT
Ch. 5 - What is the wavelength in meters of an FM radio...Ch. 5 - Conceptual APPLY 5.2 Two electromagnetic waves are...Ch. 5 - PRACTICE 5.3 The biological effects of a given...Ch. 5 - Prob. 5.4ACh. 5 - Prob. 5.5PCh. 5 - Conceptual APPLY 5.6 Compare the two elements Rb...Ch. 5 - PRACTICE 5.7 The Balmer equation can be extended...Ch. 5 - APPLY 5.8 (a) What is the longest-wavelength line...Ch. 5 - Prob. 5.9PCh. 5 - Prob. 5.10A
Ch. 5 - Give the or bital notation for an electron in an...Ch. 5 - APPLY 5.10 Extend Table 5.2 to show allowed...Ch. 5 - Conceptual PRACTICE 5.13 Give a possible...Ch. 5 - Conceptual APPLY 5.14 How many nodal planes...Ch. 5 - Give the expected ground state electron...Ch. 5 - Conceptual APPLY 5.16 Identify the atoms with the...Ch. 5 - Order the atoms from smallest to largest: Cs,...Ch. 5 - APPLY 5.18 Predict which bond length will be the...Ch. 5 - What percentage of electrical energy is converted...Ch. 5 - Prob. 5.20PCh. 5 - (a) How is the emission spectrum from the...Ch. 5 - PROBLEM 5.19 Mercury vapor is contained inside the...Ch. 5 - PROBLEM 5.20 When electricity is used to add...Ch. 5 - Atomic emission spectra arise from electron...Ch. 5 - PROBLEM 5.22 Three different wavelengths in the...Ch. 5 - Two electromagnetic waves are represented below....Ch. 5 - The following diagram shows the energy levels of...Ch. 5 - Identify each of the following orbitals, and give...Ch. 5 - Where on the blank outline of the periodic table...Ch. 5 - One of the elements shown on the following...Ch. 5 - What atom has the following orbital-filling...Ch. 5 - The following orbital-filling diagram represents...Ch. 5 - Prob. 5.33CPCh. 5 - Which has the higher frequency, red light or...Ch. 5 - Prob. 5.35SPCh. 5 - The Hubble Space Telescope detects electromagnetic...Ch. 5 - Prob. 5.37SPCh. 5 - What is the wavelength in meters of ultraviolet...Ch. 5 - What is the frequency of a microwave with...Ch. 5 - A certain cellular telephone transmits at a...Ch. 5 - Optical fibers allow the fast transmission of vast...Ch. 5 - Calculate the energies of the following waves in...Ch. 5 - The magnetic resonance imaging (MRI) body scanners...Ch. 5 - What is the wavelength in meters of photons with...Ch. 5 - What is the energy of each of the following...Ch. 5 - You’re probably familiar with using Scotch rape...Ch. 5 - Hard wintergreen-flavored candies are...Ch. 5 - Prob. 5.48SPCh. 5 - Photochromic sunglasses, which darken when exposed...Ch. 5 - Prob. 5.50SPCh. 5 - Prob. 5.51SPCh. 5 - The work function of cesium metal is 188 kJ/mol,...Ch. 5 - Prob. 5.53SPCh. 5 - The work function of silver metal is 436 kJ/mol....Ch. 5 - Cesium metal is frequently used in photoelectric...Ch. 5 - Spectroscopy is a technique that uses the...Ch. 5 - Prob. 5.57SPCh. 5 - According to the equation for the Balmer line...Ch. 5 - According to the equation for the Balmer line...Ch. 5 - Calculate the wavelength and energy in kilojoules...Ch. 5 - Calculate the wavelength and energy in kilojoules...Ch. 5 - One series of lines of the hydrogen spectrum is...Ch. 5 - One series of lines of the hydrogen spectrum is...Ch. 5 - Use the Balmer equation to calculate the...Ch. 5 - Prob. 5.65SPCh. 5 - Protons and electrons can be given very high...Ch. 5 - What is the wavelength in meters of a proton...Ch. 5 - What is the de Broglie wavelength in meters of a...Ch. 5 - What is the de Brogue wavelength in meters of a...Ch. 5 - At what speed in meters per second must a 145 g...Ch. 5 - What velocity would an electron (mass=9.111031kg)...Ch. 5 - Use the Heisenberg uncertainty principle to...Ch. 5 - The mass of a helium atom is 40026 amu, and its...Ch. 5 - What is the Heisenberg uncertainty principle, and...Ch. 5 - Why do we have to use an arbitrary value such as...Ch. 5 - What are the four quantum numbers, and what does...Ch. 5 - Tell which of the following combinations of...Ch. 5 - Give the allowable combinations of quantum numbers...Ch. 5 - Give the orbital designations of electrons with...Ch. 5 - Which of the following combinations of quantum...Ch. 5 - Which of the following combinations of quantum...Ch. 5 - What is the maximum number of electrons in an atom...Ch. 5 - What is the maximum number of electrons in an atom...Ch. 5 - Sodium atoms emit light with a wavelength of 330...Ch. 5 - Prob. 5.85SPCh. 5 - Prob. 5.86SPCh. 5 - Assign a set of four quantum numbers to each...Ch. 5 - Assign a set of four quantum numbers for the outer...Ch. 5 - Which of the following is a valid set of four...Ch. 5 - Prob. 5.90SPCh. 5 - Prob. 5.91SPCh. 5 - Prob. 5.92SPCh. 5 - Prob. 5.93SPCh. 5 - Prob. 5.94SPCh. 5 - Prob. 5.95SPCh. 5 - Order the electrons in the following orbitals...Ch. 5 - Order the following elements according to...Ch. 5 - Prob. 5.98SPCh. 5 - Prob. 5.99SPCh. 5 - Which orbital in each of the following pairs is...Ch. 5 - Order the orbitals for a multi electron atom in...Ch. 5 - According to the aufbau principle, which orbital...Ch. 5 - According to the autbau principle, which orbital...Ch. 5 - Give the expected ground-state electron...Ch. 5 - Give the expected ground-state electron...Ch. 5 - Draw orbital-filling diagrams for the following...Ch. 5 - Draw orbital-filling diagrams for atoms with the...Ch. 5 - How many unpaired electrons are present in each of...Ch. 5 - Identify the following atoms. (a) It has the...Ch. 5 - Prob. 5.110SPCh. 5 - Given the sub shells 1s, 2s, 2p, 3s, 3p and 3d,...Ch. 5 - Prob. 5.112SPCh. 5 - Prob. 5.113SPCh. 5 - Take a guess. What do you think is a likely...Ch. 5 - Take a guess. What is a likely ground-state...Ch. 5 - Prob. 5.116SPCh. 5 - Prob. 5.117SPCh. 5 - Prob. 5.118SPCh. 5 - Prob. 5.119SPCh. 5 - Prob. 5.120SPCh. 5 - Prob. 5.121SPCh. 5 - Prob. 5.122SPCh. 5 - Which atom in each of the following pairs has a...Ch. 5 - Orbital energies in single-electron atoms or ions,...Ch. 5 - Like He+ , the Li2+ ion is a single-electron...Ch. 5 - Prob. 5.126MPCh. 5 - Draw orbital-filling diagrams for the following...Ch. 5 - The atomic radii of Y (180 pm) and Li (187 pm) are...Ch. 5 - Prob. 5.129MPCh. 5 - Prob. 5.130MPCh. 5 - Prob. 5.131MPCh. 5 - The amount of energy necessary to remove an...Ch. 5 - Prob. 5.133MPCh. 5 - Prob. 5.134MPCh. 5 - Prob. 5.135MPCh. 5 - A minimum energy of 7.211019J is required to...Ch. 5 - Prob. 5.137MPCh. 5 - An energetically excited hydrogen atom has its...Ch. 5 - Prob. 5.139MP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 6.11 Define the term photon.arrow_forwardA particular microwave oven delivers 750 watts. (A watt is a unit of power, which is the joules of energy delivered, or used, per second.) If the oven uses microwave radiation of wavelength 12.6 cm, how many photons of this radiation are required to heat 1.00 g of water 1.00C, assuming that all of the photons are absorbed?arrow_forwardThe eyes of certain reptiles pass a single visual signal to the brain when the visual receptors are struck by photons of a wavelength of 850 nm. If a total energy of 3.151014 J is required to trip the signal, what is the minimum number of photons that must strike the receptor?arrow_forward
- describe waves in terms of frequency, wavelength, and amplitude.arrow_forwardGive a brief wave description of light. What are two characteristics of light waves?arrow_forwardHow does the energy possessed by an emitted photon compare to the difference in energy levels that gave rise to the emission of the photon?arrow_forward
- Given the following energy level diagram for an atom that contains an electron in the n = 3 level, answer the following questions. a Which transition of the electron will emit light of the lowest frequency? b Using only those levels depicted in the diagram, which transition of the electron would require the highest-frequency light? c If the transition from the n = 3 level to the n = 1 level emits green light, what color light is absorbed when an electron makes the transition from the n = 1 to n = 3 level?arrow_forwardA hydrogen atom in the ground stale absorbs a photon whose wavelength is 95.0 nm. The resulting excited atom then emits a photon of 1282 nm. What are the regions of the electromagnetic spectrum for the radiations involved in these transitions? What is the principal quantum number of the final state resulting from the emission from the excited atom?arrow_forwardThree emission lines involving three energy levels in an atom occur at wavelengths x, 1.5x, and 3.0x nanometers. Which wavelength corresponds to the transition from the highest to the lowest of the three energy levels?arrow_forward
- It requires 799 kJ of energy to break one mole of carbon-oxygen double bonds in carbon dioxide. What wavelength of light does this correspond to per bond? Is there any transition in the hydrogen atom that has at least this quantity of energy to one photon?arrow_forwardLight with a wavelength of 405 nm fell on a strontium surface, and electrons were ejected. If the speed of an ejected electron is 3.36 105 m/s, what energy was expended in removing the electron from the metal? Express the answer in joules (per electron) and in kilojoules per mole (of electrons).arrow_forwardWhich of the following statements is (are) true? I. The product of wavelength and frequency of light is a constant. II. As the energy of electromagnetic radiation increases, its frequency decreases. III. As the wavelength of light increases, its frequency increases. a I only b II only c III only d I and III only e II and III onlyarrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning
Quantum Mechanics - Part 1: Crash Course Physics #43; Author: CrashCourse;https://www.youtube.com/watch?v=7kb1VT0J3DE;License: Standard YouTube License, CC-BY