
Concept explainers
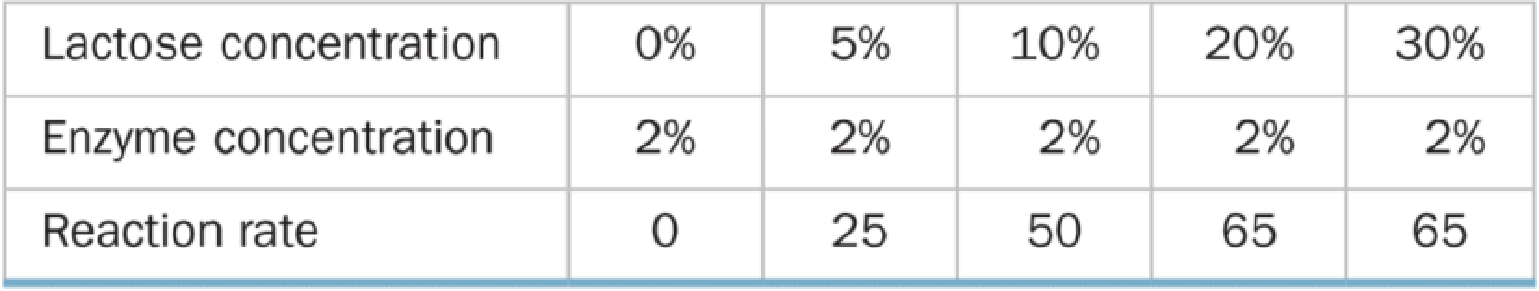
A biologist performed two series of experiments on lactase, the enzyme that hydrolyzes lactose to glucose and galactose. First, she made up 10% lactose solutions containing different concentrations of enzyme and measured the rate at which galactose was produced (grams of galactose per minute). Results of these experiments are shown in Table A below. In the second series of experiments (Table B), she prepared 2% enzyme solutions containing different concentrations of lactose and again measured the rate of galactose production.
Table A Reaction Rate and Enzyme Concentration
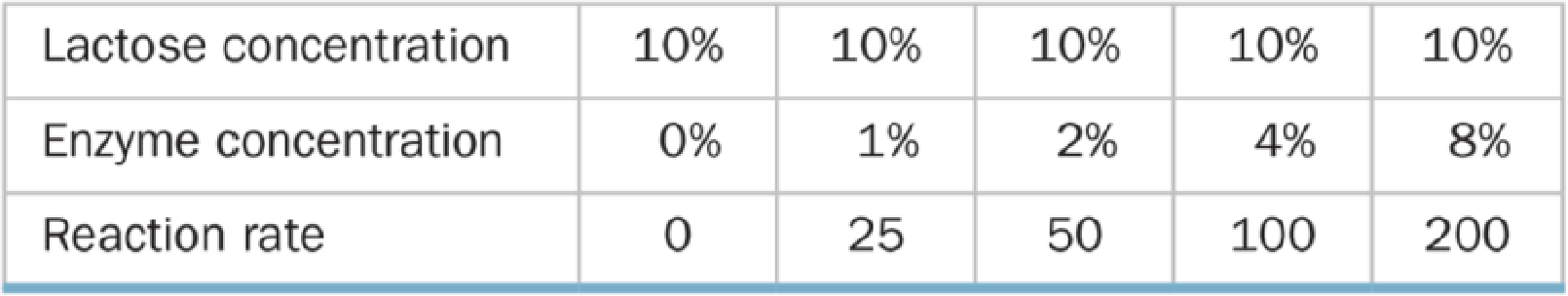
Table B Reaction Rate and Substrate Concentration
a. Graph and explain the relationship between the reaction rate and the enzyme concentration.
b. Graph and explain the relationship between the reaction rate and the substrate concentration. How and why did the results of the two experiments differ?
Trending nowThis is a popular solution!

Chapter 5 Solutions
CAMPBEL BIOLOGY:CONCEPTS & CONNECTIONS
- Idris has successfully extracted enzymatic proteins from the fish viscera (intestines and stomach). After homogenization and centrifugation, he managed to pool the crude enzyme extract. He is characterizing the enzymes. Please help Idris by answering the followingquestions:(a). How do I determine the enzyme activity? Please give the unit. (b) How do I get the specific activity of this enzyme? Please give the unit.arrow_forwardPenicillin is hydrolyzed and thereby rendered inactive by penicillinase, an enzyme present in some resistant bacteria. The mass of this enzyme in Staphylococcus aureus is 29.6 kd. The amount of penicillin hydrolyzed in 1 minute in 10 ml solution containing 10–9 g of purified penicillinase was measured as a function of the concentration of penicillin. Assume that the concentration of penicillin does not change during the assay. [Penicillin] µM Amount hydrolyzed (nanomoles) 1 0.11 3 0.25 5 0.34 10 0.45 30 0.58 50 0.61 a) Plot V0 versus [S] and 1/V0 1/[S]. Does penicillinase appear to obey Michaelis-Menten kinetics? b) What is the value of KM? c) What is the value of Vmax? d) What is the turnover number of penicillinase under these conditions?arrow_forwardIdris has successfully extracted enzymatic proteins from the fish viscera (intestines and stomach). After homogenization and centrifugation, he managed to pool the crude enzyme extract. He is characterizing the enzymes. Please help Idris by answering the following questions:(a) How do I determine the protein/enzyme concentration? Please give the unit.arrow_forward
- a. Which unknown sample is the negative control? And Which unknown sample is the positive control? b. The experiment she conducted aimed to determine the action of what enzyme?arrow_forwardFor each of the four possibilities listed below (a through d), only one answer is correct and the others are false. Identify the correct statement and explain why the others are false. In your answer, write CORRECT next to the statement that you think is true, and ONLY for the ones that you think are false, explain what is wrong with the statement . When glucose reacts with ATP to form glucose-6-phosphate (as shown on the figure below): 1. The synthesis of glucose-6-phosphate is exergonic 2. ADP is at a higher energy level than ATP 3. Glucose-6-phosphate is at a higher energy level than glucose 4. Because ATP donates a phosphate to glucose, this is not a coupled reactionarrow_forwardTyrosinase enzyme activity is assayed by monitoring the oxidation of 3, 4-dihydroxyphenylalanine (dopa) to the red-colored dopachrome. Calculate the tyrosinase activity (U/mL) by using the experimental data given belowarrow_forward
- Refer to the figure below and answer the following questions: Legend: Blue – wild-type β-galactosidase; Red – mutant β-galactosidase _________ a. What is the optimum pH of wild type β-galactosidase? _________ b. What is the optimum temperature of mutant β-galactosidase? _________ c. Which enzyme has the greater activity at pH 7.2? _________ d. Which enzyme has the greater activity at a temperature of 42.5oC? _________ e. Which enzyme has greater activity if pH decreases from 7.5 to 6.4? _________ f. Which enzyme has greater activity if temperature increases from 40oC to 41 oC?arrow_forwardA newly developed drug is suspected to act by inhibiting its enzymatic target. To test this, the rates of reaction in the presence and absence of the new drug were determined. The results of these experiments have been plotted as a double reciprocal plot shown in figure 1 below. Using this figure, calculate the values of Vmax and Km for the enzyme in the presence and absence of the drug. Based on the figure and your answer, what type of inhibition is the new drug displaying? Briefly explain how this type of inhibition exerts its action on an enzyme.arrow_forwardYou design an experiment to explore the effects of enzyme concentration on the rate of glucose (product) production. In the experiment, you prepare 5 tubes with equal amounts of water and substrate but altered the amount of enzyme. Tube #1 did not have enzyme, Tube #2 had 100 mg of enzyme, Tube #3 had 200 mg of enzyme, Tube #4 had 300 mg of enzyme, Tube #5 had 400 mg of enzyme. The results are summarized in the graph. Select all that apply. Given enough time all tubes (#1-5) would reach 1100 mg/dL. The substrate in tube #1 was denatured at 40 minutes. The fastest rate of reaction was in Tube #4. Adding more enzyme to Tube #5 at 40 minutes would produce more product. Tube #4 and #5 have run out of substrate at 40 minutes.arrow_forward
- Pyridoxal phosphate (PLP) is a coenzyme for the enzyme ornithine aminotransferase. The enzyme was purified from cells grow in PLP = deficient media as well as from cells grown in media that contained pyridoxal phosphate. The stability of the two different enzyme preparations was then measured by incubating the enzyme at 37°C for different lengths of time and then assaying for the amount of enzyme activity remaining. The following results were obtained. (a) Why does the amount of active enzyme decrease with the time of incubation? (b) Why does the amount of enzyme from the PLP deficient cells decline more rapidly?arrow_forwardIdris has successfully extracted enzymatic proteins from the fish viscera (intestines and stomach). After homogenization and centrifugation, he managed to pool the crude enzyme extract. He is characterizing the enzymes. Please help Idris by answering the followingquestions:arrow_forwardCompounds I, II, and III are in the following biochemical pathway: precursor compound I enzyme A enzyme B compound II ► compound III enzyme Mutation a inactivates enzyme A, mutation b inactivates enzyme B, and mutation c inactivates enzyme C. Mutants, each having one of these defects, were tested on minimal medium to which compound I, II, or III was added. Fill in the results expected of these tests by placing a plus sign (+) for growth or a minus sign (-) for no growth in the table below. Minimal medium to which is added Strain with mutation Compound I Compound II Compound IIIarrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





