
SCIENTIFIC THINKING Mercury is known to inhibit the permeability of water channels. To help establish that the protein isolated by Agre’s group was a water channel (see Module 5.7), the researchers incubated groups of RNA-injected oocytes (which thus made aquaporin proteins) in four different solutions: plain buffer, low concentration and high concentration of a mercury chloride (HgCl2) solution, and low concentration of a mercury solution followed by an agent (ME) known to reverse the effects of mercury. The water permeability of the cells was determined by the rate of their osmotic swelling. Interpret the results of this experiment, which are presented in the graph below.
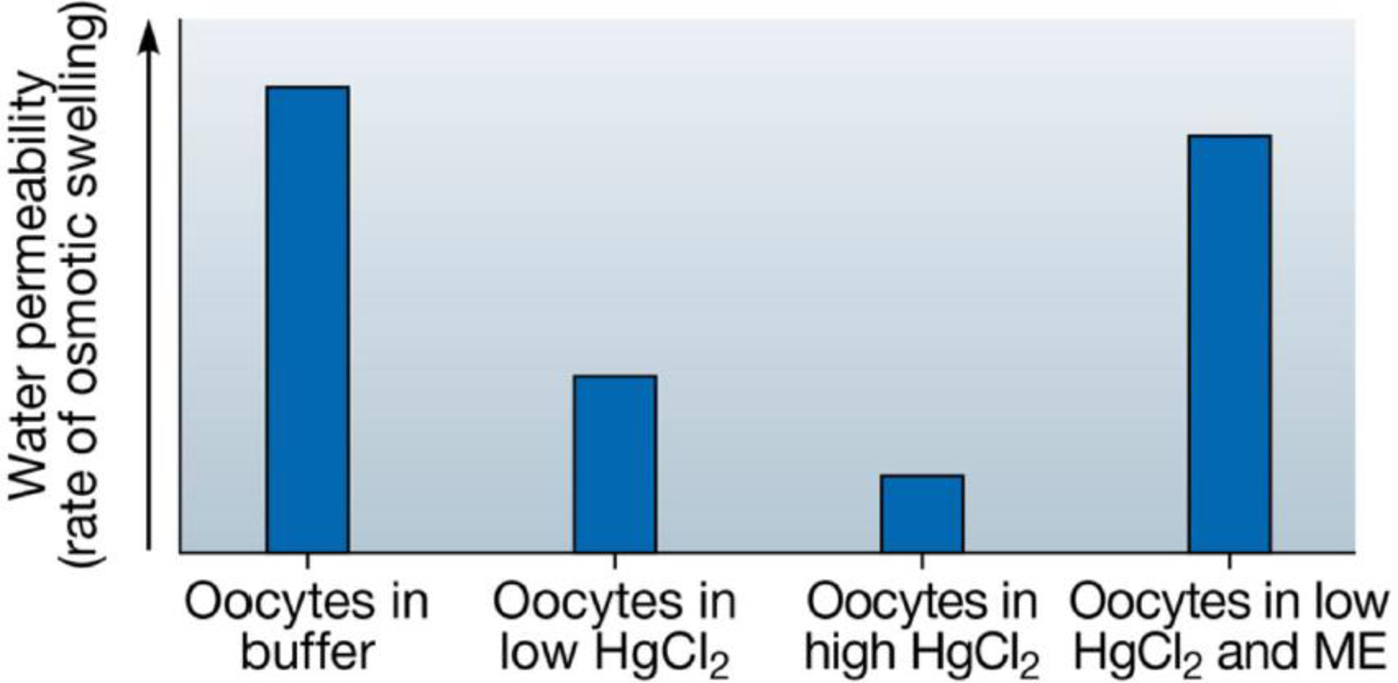
Data from G. M. Preston et al., Appearance of water channels in Xenopus oocytes expressing red cell CHIP28 protein, Science 256: 3385–7 (1992).
Control oocytes not injected with aquaporin RNA were also incubated with buffer and the two concentrations of mercury. Predict what the results of these treatments would be.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
CAMPBEL BIOLOGY:CONCEPTS & CONNECTIONS
- In an investigation of osmosis, apple cores were submerged in different molarity solutions to determine the unknown osmolarity of the apple. To determine the molarity five uniform apple cores were massed before submerging them in different molarity solutions. After 24 hours the apple cores were massed again. Then the percent change in mass was determined for the five uniform apple cores in each molarity solution. (b) Identify the osmolarity of the apple.arrow_forwardCompound Sodium Lactate is isotonic solution. this mean that it a. reduces movement of sodium across the membrane b.prevent movement of water across the semi permerable cell membrane c. promotes movement of water out of the cell d. facilitates equal movement of water across the semi permerable cell membranearrow_forwardyeasts are able to produce high internal concentrations of glycerol to counteract the osmotic pressure of the surrounding media. suppose that a sample of yeast cells were placed in a 4% sodium chloride solution by weight. The density of solution is at 25 C = 1.02 g/ml, Molecular weight of solute = 58.44 g/mol, i of glycerol = 1 and R=0.08205 L-atm/mol-K What is the weight of solute in grams What is the moles of solute What is the volume of the solution in liters What is the molarity of the solution What is the value of the temperature to be used to solved for the osmotic pressure of the solution What is the osmotic pressure of solutionarrow_forward
- Scientists let rice blast fungal infection cells attach to and attempt to penetrate six different polymer sheets for different lengths of time and in varying extracellular osmotic pressures (Figure 22.9). As the number of the sheet increases so does its hardness. Which of the following makes the most sense based on the data? 100- 60 40- 20- 192 incubation time (hr) 72 96 240 100- 80- 60- 40- 20- b-Oo 8 10 12 extracellular osmotic pressure, MPa Figure 22.9 Penetration as a function of incubation time (A) and extracellular osmotic pressure (B). Numbers 1 to 6 in both panels indicate the substrate, with lower numbers being softer substrates. Percent penetration goes down over time and as substrate hardness increases. If penetration of a substrate occurs at a particular external pressure, this indicates that internal pressure is high enough to allow penetration of the particular substrate. Harder substrates are more easily penetrated when extracellular osmotic pressures are low. The…arrow_forwardIdentify the movement of water in surrounding medium and intracellular environment, given these hypothetical conditions. a. surrounding medium (7% salt, 93 % water) b. intracellular environment - RBC (25% salt, 75% water) 2. a. surrounding medium (35% salt, 65% water) b. intracellular concentration - RBC ( 2% salt, 98% water)arrow_forwardIn the experiment showing osmosis through a semipermeable membrane, 3 corn syrup mixtures are used. If equal volumes of two solutions (10% corn syrup and 30% corn syrup) were placed in two beakers that were separated by a semipermeable membrane, permeable only to water, and let sit for 1 hour, what would be the expected concentration/s of the two solutions at the end of the time period? Explain using solute concentration, water concentration etc.arrow_forward
- Osmosis and Red Blood Cells Imagine you are working in a research lab studying red blood cells. You prepare 5 mL solutions of salt dissolved in pure water at 0, 0.9, 1.5, and 5% NaCl. You have another solution of red blood cells, and you gently mix it and then add 100 uL of it to each of the three 5 mL solutions. You gently mix and observe a sample of each under the microscope. Results: Red blood cells look intact and normal in the 0.9% solution. In the 1.5% solution cells are present but look shriveled. In the 0 and 5% NaCl solutions, you do not see any cells at all! Exercise #2: Explain the results. Exercise #3: Would you expect to see the same effect as above on plant cells within intact plant tissue? Explain your answer.arrow_forwardFor a lab on diffusion and osmosis, where a egg yolk was placed in a cup water the following question asks Consider a scenario in which the size of an egg yolk remains unchanged after in water soaking for an hour. What are two possible explanations as to why this occurs? What would two reasons be for the yolk to be unchanged after a hour in water?arrow_forwardIn an experiment, a 0.001 (mole fraction) solution of polysaccharide in water is made and is placed in the compartment A. Compartment B is filled with pure water. The two compartments are separated by a porous semi-permeable membrane that allows the exchange of water molecules between the two compartments, but not that of the larger polysaccharide molecules. part 1: Show that the chemical potential of water in compartment A is lower than that in compartment B by 2.48 J/mol.arrow_forward
- In medicine, why is it important to administer only isotonic intravenous solutions to patients? Example of isotonic IV fluids are normal saline and lactated Ringer’s solution.arrow_forwardDiffusion experiments of a small molecule drug in tissue samples are performed. The drug has a radius of 2.24 nm and a pure-water diffusivity of 2.75 x 10^-6 cm^2/s. Cells occupy approximately 72% of the tissue byvolume. The molecule is very hydrophilic and does not readily cross cellular membranes. If you assume that the interstitial space is gel-like with a microstructure parameter k=3.7 x 10^-3 nm^-1, what is the effective diffusivity in the tissue?arrow_forwardMelvin, ace biology student, is making a display for Ms. Fishhawk's class to show what "isotonic," "hypertonic" and "hypotonic" mean. Melvin has a tank where the two sides are separated by a semi-permeable membrane. The figure shows the solutions that Melvin pours into each side of the tank. 30% starch 70% water A 5% starch 95% water B the a. Label the figure to show which side is the hypertonic side and which is the hypotonic side. b. After loading the tank with the solutions, Melvin lets the tank sit for 20 minutes. Assuming membrane is the same as the dialysis membranes we used in lab, will the volume of the two solutions change? In what way?arrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





