
Concept explainers
Using the Fischer projection of
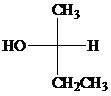
Switching the positions of
Switching the positions of
Switching the positions of three groups.
Switching
Rotating the Fischer projection
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Organic Chemistry - Standalone book
- Consider CH3-CH(OH)-CH(OH)(Br). a.How many stereogeniccenters are in the molecule? b.How many stereoisomers are there for the compound? c.Draw the Fischer projection for each of the stereoisomer. Label each using I, II, etc. d.Which pairs are enantiomers? Which are diastereomers? e.Determine the absolute configuration of each chiral center in one pair of diastereomer.arrow_forward1. Locate/Label the chirality center of the given molecules. a. OH H2N C H CH3 CH3 SH b. OH HO CH2NHC(CH3)3 HOCH2arrow_forwardWrite the IUPAC names for each compound showing the R or S configuration of each chiral center.arrow_forward
- HO OH a) Provide the Fischer projection of the above molecule. b) provide the R-S configuration for each of the chiral centre(s) c) Draw the Fischer projection and the bond line structure of the above molecule's enantiomerarrow_forwardWhat is the configuration at the chiral centers of the Fischer projection of the molecule shown? (The molecule is drawn with C1 at the top and C5 at the bottom.)arrow_forwardDraw a Fischer projection of (2R,3S)-3-bromo-2-butanolarrow_forward
- Assign R or S configuration to each chirality center in the following molecules. First assign the configuration of the chirality center closest to the top of the Fischer projection. Then give the configuration of the other chirality center. CH₂OH -OH CH3CH₂ # H- -OCH3 CO₂H 1st structure: Top chirality center 2nd structure: Top chirality center H3C- H3C- OH -OH -H Other chirality center: ✓. Other chirality center: Varrow_forward8. Assign each chiral carbon as R or S in compound-A. Draw the mirror image of compound-A and assign configuration to each chiral carbon. (note that the dotted line represents the mirror). H₂N S OH Compound A CH3 CH3 mirror CH3 CH 3 Mmok NHZarrow_forward4. Determine if each Fischer projection is chiral or achiral. If chiral, identify it as the d or l stereoisomer and draw the mirror image. a. CH₂OH HO- ti CH3 H b. CH ₂ 0+H Н CH3 HO ₂-CH3arrow_forward
- Consider the below structure: ОН 1 H3CHN a. Draw 3-D structures and a Fischer projection for the 1R, 2S, and the 1S, 2S stereoisomers of the above structure. b. Draw all the conformers obtained by rotation about the C1-C2 bond and rank them in order of increasing stability.arrow_forward1. Circle the chiral centers in this compound and draw a Fischer projection. CH--CH2 ot OH 2. Number the carbon atoms in the molecule shown and draw the ring structure for this compound, using the -OH group on C#5. Circle the hemiacetal functional group. Number the carbon atoms in the ring structure. CHOH c=O -OH Ho Ht molecle presented tn the drawing be CHOH dentiy the hr onal ro cley onim 3. Number the carbon atoms in the monosaccharides shown and draw structures to show how a (1→ 6) glycosidic linkage will form. Circle the glycosidic linkage. What type of functional group is this? CH2OH OH OH a-glucosearrow_forward2. Answer the following: a) b) Identify the stereogenic center by placing an asterisk and determine the configuration of each. What's the relationship between the following pairs of Fischer Projection structures? Same, Enantiomers or Diastereomers? (Note: make sure to review the posted Fischer Projection Rules for this question) HO HO H H CH3 H H CH₂CH3 CH 3 CH3 -OH H HOCH 3 CH₂CH3 H- HO OH H CH3 H CH 3arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
