
Concept explainers
(a)
Interpretation: To determine the type of reaction whether it will be an
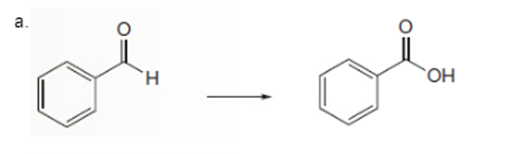
Concept Introduction: Oxidation, which refers to the loss of electrons, is the increase in the oxidation state of its component atoms. When an atom obtains electrons or has its oxidation state reduced, the reduction can occur.
(b)
Interpretation: To determine the type of reaction whether it will be an oxidation or reduction reaction.
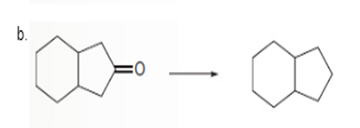
Concept Introduction:
Oxidation, which refers to the loss of electrons, is the increase in the oxidation state of its component atoms. When an atom obtains electrons or has its oxidation state reduced, the reduction can occur.
(c)
Interpretation: To determine the type of reaction whether it will be an oxidation or reduction reaction.
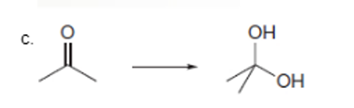
Concept Introduction: Oxidation, which refers to the loss of electrons, is the increase in the oxidation state of its component atoms. When an atom obtains electrons or has its oxidation state reduced, the reduction can occur.
(d)
Interpretation: To determine the type of reaction whether it will be an oxidation or reduction reaction.
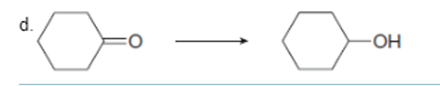
Concept Introduction: Oxidation, which refers to the loss of electrons, is the increase in the oxidation state of its component atoms. When an atom obtains electrons or has its oxidation state reduced, the reduction can occur.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
ORGANIC CHEMISTRY (LOOSELEAF)
- The conversion of reactant R to product A is under control and is preferred at temperature kinetic, low kinetic, high thermodynamic, low thermodynamic, high The conversion of reactant R to product B is under control and is preferred at temperature kinetic, low kinetic, high thermodynamic, low thermodynamic, higharrow_forwardDesribe the reaction product.arrow_forwardPlease indicate an arrow push for each reaction.arrow_forward
- Draw an energy diagram for each reaction in Problem 20.28, paying attention to the relative energies of the overall reactants, overall products, and any intermediates.arrow_forward:Draw two major product (stereoisomers) : Reaction type for the major product :Draw all the minor products (identify # of products) :Reaction type for the minor product(s)arrow_forwardThis reactions is a ? Oxidation Reduction Rearrangement Hydrolysis Dehydration/condensationarrow_forward
- Draw a detailed free energy diagram for the following reaction. Include and label the overall reactants, the overall products, the intermediate(s), the axis, and the transition state(s). 0 NaOCH₂CH3 CI OCH₂CH3+ NaClarrow_forwardDraw the products of each combustion reaction.arrow_forwardThe rate determining step of a reaction is the slowest step and thus, has the lowest energy barrier first step on a staircase fastest step and thus, has the lowest energy barrier slowest step and thus, has the highest energy barrier fastest step and thus, has the highest energy barrierarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





