
ORGANIC CHEMISTRY (LOOSELEAF)
6th Edition
ISBN: 9781260475630
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4, Problem 78P
Read Appendix B on naming bicyclic compounds. Then give IUPAC name for each of the following compounds.
a. 
b. 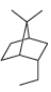
c. 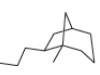
d. 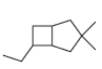
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Provide the two isomers of compound A. Please provide the IUPAC name for each.
1st isomer: has quarternary, secondary, and primary carbons
2nd isomer: has secondary and primary carbons
Provide the IUPAC name for the compound.
A.
Br.
В.
NH2
C.
D.
OH
1) Write the IUPAC name for each molecule below.
a.
b.
C.
HN
N.
Chapter 4 Solutions
ORGANIC CHEMISTRY (LOOSELEAF)
Ch. 4.1 - Prob. 1PCh. 4.1 - Problem 4.2 Which of the following is not another...Ch. 4.1 - Problem 4.3 Draw the five constitutional isomers...Ch. 4.1 - Prob. 4PCh. 4.1 - Prob. 5PCh. 4.2 - Draw the five constitutional isomers that have...Ch. 4.4 - Problem 4.7 Give the IUPAC name for each...Ch. 4.4 - Give the IUPAC name for each compound. a....Ch. 4.4 - Problem 4.9 Give the structure corresponding to...Ch. 4.4 - Prob. 10P
Ch. 4.5 - Give the IUPAC name for each compound.Ch. 4.5 - Give the structure corresponding to each IUPAC...Ch. 4.8 - Arrange the following compounds in order of...Ch. 4.9 - Problem 4.14 Draw the staggered and eclipsed...Ch. 4.9 - Prob. 15PCh. 4.9 - Prob. 16PCh. 4.10 - Problem 4.17 a. Draw the three staggered and...Ch. 4.10 - Problem 4.18 Rank the following conformations in...Ch. 4.10 - Problem 4.19 Consider rotation around the...Ch. 4.10 - Calculate the destabilization present in each...Ch. 4.12 - Problem 4.21 Classify the ring carbons as up or...Ch. 4.12 - Problem 4.22 Using the cyclohexane with the C’s...Ch. 4.13 - Draw a second chair conformation for each...Ch. 4.13 - Problem 4.24 Draw both conformations for and...Ch. 4.13 - Problem 4.25 Draw the structure for each compound...Ch. 4.13 - Prob. 26PCh. 4.14 - Prob. 31PCh. 4.14 - Prob. 32PCh. 4.15 - Prob. 33PCh. 4 - Name each alkane using the ball-and-stick model,...Ch. 4 -
4.40 Draw the structure corresponding to each...Ch. 4 - 4.42 Give the IUPAC name for each compound.
a....Ch. 4 - Prob. 42PCh. 4 - 4.46 Considering rotation around the bond...Ch. 4 - 4.50 Calculate the barrier to rotation for each...Ch. 4 - 4.51 The eclipsed conformation of is less...Ch. 4 - (a) Draw the anti and gauche conformations for...Ch. 4 - For each compound drawn below: a.Label each OH,Br...Ch. 4 - Draw the two possible chair conformations for...Ch. 4 - For each compound drawn below: a. Draw...Ch. 4 - Classify each pair of compounds as constitutional...Ch. 4 - Prob. 66PCh. 4 - 4.64 Draw the products of combustion of each...Ch. 4 - 4.65 Hydrocarbons like benzene are metabolized in...Ch. 4 - Prob. 69PCh. 4 - Prob. 70PCh. 4 - Cyclopropane and cyclobutane have similar strain...Ch. 4 - Prob. 72PCh. 4 - Haloethanes (CH3CH2X,X=Cl,Br,I) have similar...Ch. 4 - Prob. 74PCh. 4 - Prob. 75PCh. 4 - Consider the tricyclic structure B (a) Label each...Ch. 4 - Read Appendix B on naming branched alkyl...Ch. 4 - Read Appendix B on naming bicyclic compounds. Then...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Consider a sample of ideal gas initially in a volume V at temperature T and pressure P. Does the entropy of thi...
General Chemistry: Principles and Modern Applications (11th Edition)
Fully developed conditions are known to exist for water flowing through a 25-nim-diameer tube at 0.01 kg/s and ...
Fundamentals of Heat and Mass Transfer
Classify each example of molecular art as a pure element, a pure compound, or a mixture.
General, Organic, and Biological Chemistry - 4th edition
4.1 Write the symbols for the following elements.
a. copper
b. platinum
c. calcium
d. manganese
e. Iron
...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 1. Provide the IUPAC name for each of the following organic molecules. a. b. C. H3C H3C, Home H3C CH3 CH3 CH3 Br H 2. Draw structures for the following organic molecules, using the wedged/hashed line convention to depict any stereochemical relationships between groups: a. cis-3-ethylcyclobutane-1-carbaldehyde b. (R)-1-chloro-6-iodo-6-methyloctan-3-onearrow_forwardWhich heteroatom don't contain heterocyclic compounds? a. Phosphorus b. Oxygen c. Nitrogen d. Sulfurarrow_forwardRank the following compounds in order from smallest heat of combustion to largest heat of combustion. A Barrow_forward
- Which compound is second least stable, giving off the second most heat of hydrogenation or combustion (when burned)? a. O b. d. Of.arrow_forwardProvide the IUPAC name for the following. You may use either the 'prefix-2-alkene' or the 'prefixalk-2-ene' naming convention, but don't forget to include any stereoisomerism in your name if relevant. a. C. Br CH₂ b. farrow_forwardGive the IUPAC names according to the rulesarrow_forward
- 1. Give the IUPAC name for the provided structures a. thy equ b. d. CIarrow_forwardDraw the structure corresponding to each IUPAC name. a. 2,3,4,5−tetramethyldecane draw structure ... b. cyclononane draw structure ...arrow_forward1. What is the relationship between the following two compounds?a.identical b.not isomers; different compounds entirely c.constitutional isomers2. Is the following alkene cis, trans, or neither?arrow_forward
- A. Name the following compounds according to IUPAC rulesarrow_forwardHydrocarbons like benzene are metabolized in the body to arene oxides, which rearrange to form phenols. This is an example of a general process in the body, in which an unwanted compound (benzene) is converted to a more water-soluble derivative called a metabolite, so that it can be excreted more readily from the body. a.) Classify each of these reactions as oxidation, reduction, or neither.b.) Explain why phenol is more water soluble than benzene. This means that phenol dissolves in urine, which is largely water, to a greater extent than benzenearrow_forwardGive the correct IUPAC name and structure of the ff.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License