
Interpretation: Structure properties of covalent compounds that can dissociate into ions if it dissolves in water should be determined. Type of solution formed by these solutions should be determined. Also, three examples for same type of solution should be determined.
Concept introduction: Solution is made by two portions, solute and solvent. Solute represents substance present in small quantity while solvent represents substance that dissolves solute.
Covalent compounds are formed from sharing of electrons between the non-metals. These covalent compounds are classified into polar covalent compounds and nonpolar covalent compounds.
Polarity in molecule is generated if it has unequal distributed electron density between bonds. Polar molecule has partial charges on atoms of the molecule. The general representation of polar molecule is as follows:
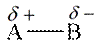
Here, B has high electronegative value than A
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Principles of General Chemistry
- What mass of a 4.00% NaOH solution by mass contains 15.0 g of NaOH?arrow_forwardMaple syrup sap is 3% sugar (sucrose) and 97% water bymass. Maple syrup is produced by heating the sap toevaporate a certain amount of the water. (a) Describe what happens to the composition and boilingpoint of the solution as evaporation takes place. (b) A rule of thumb among maple syrup producers is thatthe finished syrup should boil about 4 C higher than theoriginal sap being boiled. Explain the chemistry behindthis guideline. (c) If the finished product boils 4 C higher than the originalsap, calculate the concentration of sugar in the finalproduct. Assume that sugar is the only solute and theoperation is done at 1 atm pressure.arrow_forwardDissolving 3.0 g of CaCl2(s) in 150.0 g of water in a calorimeter (Figure 5.12) at 22.4 °C causes the temperature to rise to 25.8 °C. What is the approximate amount of heat involved in the dissolution, assuming the specific heat of the resulting solution is 4.18 J/g °C? Is the reaction exothermic or endothermic?arrow_forward
- Without consulting your textbook, list and explain the main postulates of the kinetic molecular theory for gases. How do these postulates help us account for the following bulk properties of a gas: the pressure of the gas and why the pressure of the gas increases with increased temperature; the fact that a gas tills its entire container; and the fact that the volume of a given sample of gas increases as its temperature is increased.arrow_forwardClassify each of the following solutions as saturated, unsaturated, or supersaturated based on the following observations made after adding a small piece of solid solute to the solution. a. The added solute rapidly dissolves. b. The added solute falls to the bottom of the container where it remains without any decrease in size. c. The added solute falls to the bottom of the container where it decreases in size for several hours and thereafter its size remains constant. d. The added solute causes the production of a large amount of solid white crystals.arrow_forwardThe names and chemical formulae of some chemical compounds are written in the first two columns of the table below. Each compound is soluble in water. Imagine that a few tenths of a mole of each compound is dissolved in a iter of water. Then, write down in the third column of the table the chemical formula of the major chemical species that will be present in this solution. For example, you know water itself will be present, so you can begin each list with the chemical formula for water (H,O). Note: "major" chemical speces are those present in concentrations greater than 10° molL. major species present when dissolved in water compound formula GH(OH), propvlene vcol sopropand vitreidearrow_forward
- The names and chemical formulae of some chemical compounds are written in the first two columns of the table below. Each compound is soluble water. Imagine that a few tenths of a mole of each compound is dissolved in a liter of water. Then, write down in the third column of the table the chemical formula of the major chemical species that will be present in this solution. For example, you know water itself will be present, so you can begin each list with the chemical formula for water (H₂O). -6 Note: "major" chemical species are those present in concentrations greater than 10 compound isopropanol zinc chloride glycerol formula C3H8O ZnCl₂ C₂H₂O3 major species present when dissolved in water 0 0 0 mol/L. 0,0,...arrow_forwardAdding food coloring to water creates a solution. Which of these is true of all solutions? They all include water. They all include water. The parts are evenly mixed. The parts are evenly mixed. They are all liquids or gases. They are all liquids or gases. The substances are permanently combined. The substances are permanently combined.arrow_forwardThe names and chemical formulae of some chemical compounds are written in the first two columns of the table below. Each compound is soluble in water. Imagine that a few tenths of a mole of each compound is dissolved in a liter of water. Then, write down in the third column of the table the chemical formula of the major chemical species that will be present in this solution. For example, you know water itself will be present, so you can begin each list with the chemical formula for water (H₂O). -6 Note: "major" chemical species are those present in concentrations greater than 10 mol/L. compound acetone iron (III) chloride zinc bromide formula (CH,),CO FeCl3 Zn Br₂ major species present when dissolved in water 0 0 X 0,0,... Śarrow_forward
- 8, Q2arrow_forwardThe names and chemical formulae of some chemical compounds are written in the first two columns of the table below. Each compound is soluble in water. Imagine that a few tenths of a mole of each compound is dissolved in a liter of water. Then, write down in the third column of the table the chemical formula of the major chemical species that will be present in this solution. For example, you know water itself will be present, so you can begin each list with the chemical formula for water (H₂O). Note: "major" chemical species are those present in concentrations greater than 10 mol/L. compound methanol fructose iron(II) iodide formula CH₂OH C6H12O6 FeL₂ major species present when dissolved in water 0 X 0,0,... Śarrow_forwardThe names and chemical formulae of some chemical compounds are written in the first two columns of the table below. Each compound is soluble in water. Imagine that a few tenths of a mole of each compound is dissolved in a liter of water. Then, write down in the third column of the table the chemical formula of the major chemical species that will be present in this solution. For example, you know water itself will be present, so you can begin each list with the chemical formula for water (H,O). -6 Note: "major" chemical species are those present in concentrations greater than 10 mol/L. major species present when dissolved in water compound formula 0,0,.. nickel(II) chloride NiCl, sodium nitrate NaNO3 nickel(II) iodide Nil,arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStaxChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStaxChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co





